Antifungal Drugs
Since the 20th century, people have successively developed effective anti-fungal drugs such as griseofulvin. Flucytosine had been first introduced in the 1960s and its anti-fungal spectrum is limited to Candida and Cryptococcus, and is only used in combination with amphotericin B (AmB). A new era of the antifungal treatment is the development of the azole drug which reduces the toxicity and improves the antifungal effectiveness. The first drug, clotrimazole, was used only for the treatment of candidiasis infection of local mucosal and skin; the second, miconazole, is currently used only locally. It is not until the mid of 1970s when ketoconazole has been successfully developed before oral administration become effective against deep fungal infection. Ketoconazole has broad anti-bacterial spectrum and reduced toxicity, being considered to be a revolution during the development of antifungal drugs. However, severe patients can’t subject to oral administration for absorption. Moreover, the poor central nervous system penetration capability and poor antifungal activity as well as severe liver toxicity are limiting its clinical use.
In the late 1980s, people had successively developed itraconazole and fluconazole with more remarkable improved antimicrobial spectrum and bioavailability. Their emergence marked a new era of antifungal therapy. Even so, there are still no drugs that are absolutely fungicidal. In recent years, with increasing number of resistant strains, coupled with the limitations of drug toxicity, the treatment against fungal infection is facing many challenges. Currently the relatively promising drugs are drugs inhibiting the membrane ergosterol synthesis and interfering with cell wall 0-glucan synthesis, such as fluconazole, itraconazole and ciliary eumycetin. In addition, those old drugs with strong antibacterial effect but large toxicity are also subjecting to continuous improvement. Combination of two or more drugs is also the direction of antifungal therapy.
- Structure:
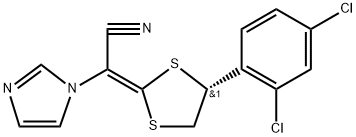
- Chemical Name:Luliconazole
- CAS:187164-19-8
- MF:C14H9Cl2N3S2
- Structure:
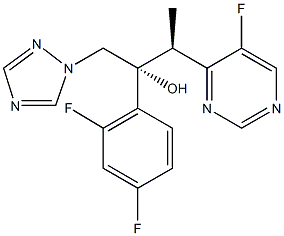
- Chemical Name:Voriconazole
- CAS:137234-62-9
- MF:C16H14F3N5O
- Structure:
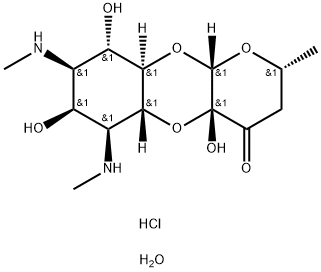
- Chemical Name:Spectinomycin dihydrochloride pentahydrate
- CAS:22189-32-8
- MF:C14H27ClN2O8
- Structure:
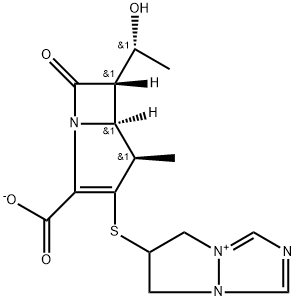
- Chemical Name:Biapenem
- CAS:120410-24-4
- MF:C15H18N4O4S
- Structure:
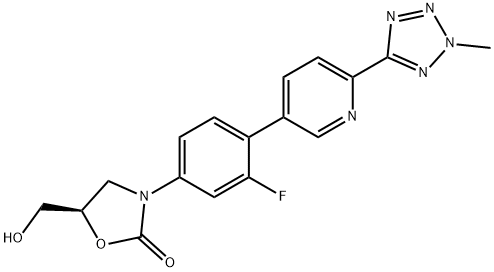
- Chemical Name:Torezolid
- CAS:856866-72-3
- MF:C17H15FN6O3
- Structure:
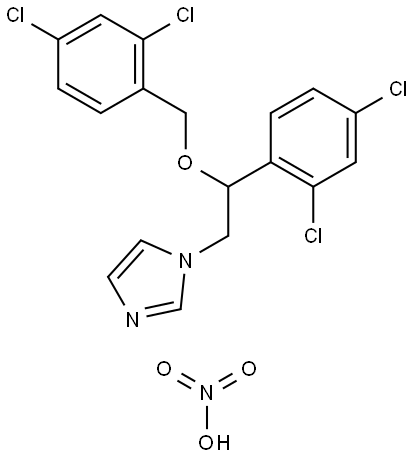
- Chemical Name:Miconazole nitrate
- CAS:22832-87-7
- MF:C18H14Cl4N2O.HNO3
- Structure:
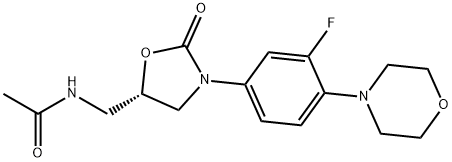
- Chemical Name:Linezolid
- CAS:165800-03-3
- MF:C16H20FN3O4
- Structure:
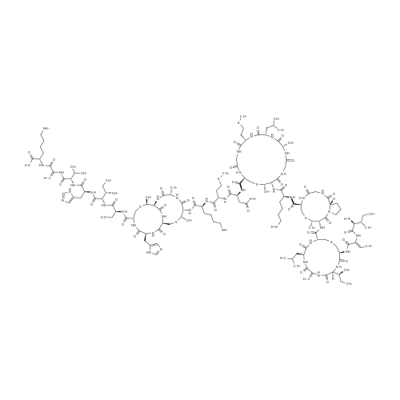
- Chemical Name:Nisin
- CAS:1414-45-5
- MF:C143H230N42O37S7
- Structure:
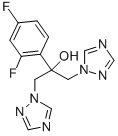
- Chemical Name:Fluconazole
- CAS:86386-73-4
- MF:C13H12F2N6O
- Structure:
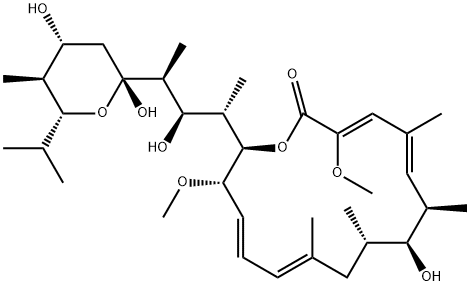
- Chemical Name:BAFILOMYCIN A1
- CAS:88899-55-2
- MF:C35H58O9
- Structure:
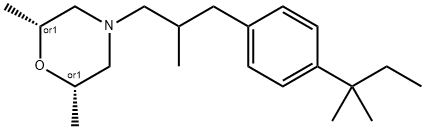
- Chemical Name:Amorolfine
- CAS:78613-35-1
- MF:C21H35NO
- Structure:
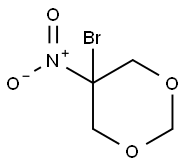
- Chemical Name:5-Bromo-5-nitro-1,3-dioxane
- CAS:30007-47-7
- MF:C4H6BrNO4
- Structure:
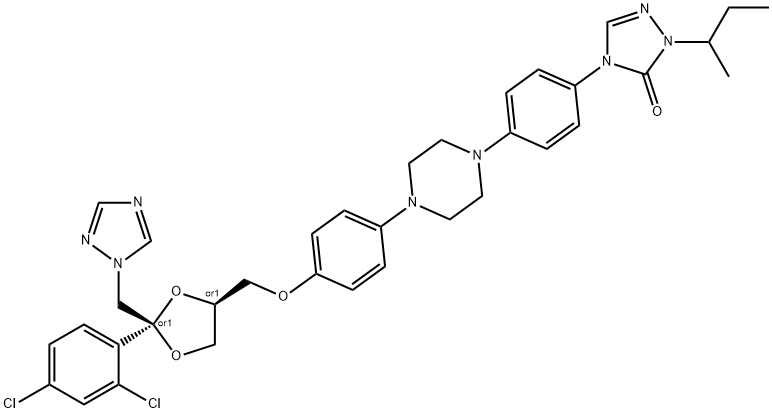
- Chemical Name:Itraconazole
- CAS:84625-61-6
- MF:C35H38Cl2N8O4
- Structure:
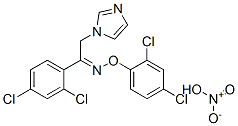
- Chemical Name:Oxiconazole nitrate
- CAS:64211-46-7
- MF:C18H13Cl4N3O
- Structure:
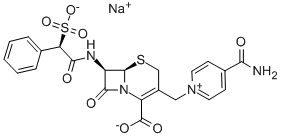
- Chemical Name:cefsulodin sodium salt
- CAS:52152-93-9
- MF:C22H19N4O8S2.Na
- Structure:
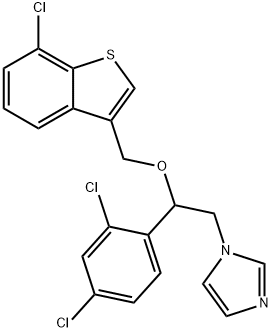
- Chemical Name:Sertaconazole
- CAS:99592-32-2
- MF:C20H15Cl3N2OS
- Structure:
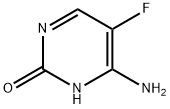
- Chemical Name:5-Fluorocytosine
- CAS:2022-85-7
- MF:C4H4FN3O
- Structure:
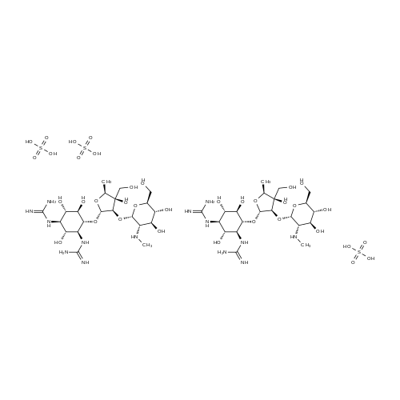
- Chemical Name:Dihydrostreptomycin sulfate
- CAS:5490-27-7
- MF:C21H43N7O16S
- Structure:
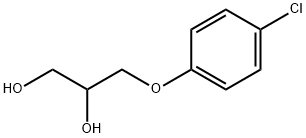
- Chemical Name:Chlorphenesin
- CAS:104-29-0
- MF:C9H11ClO3
- Structure:
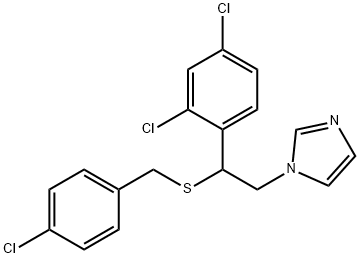
- Chemical Name:SULCONAZOLE
- CAS:61318-90-9
- MF:C18H15Cl3N2S
- Structure:
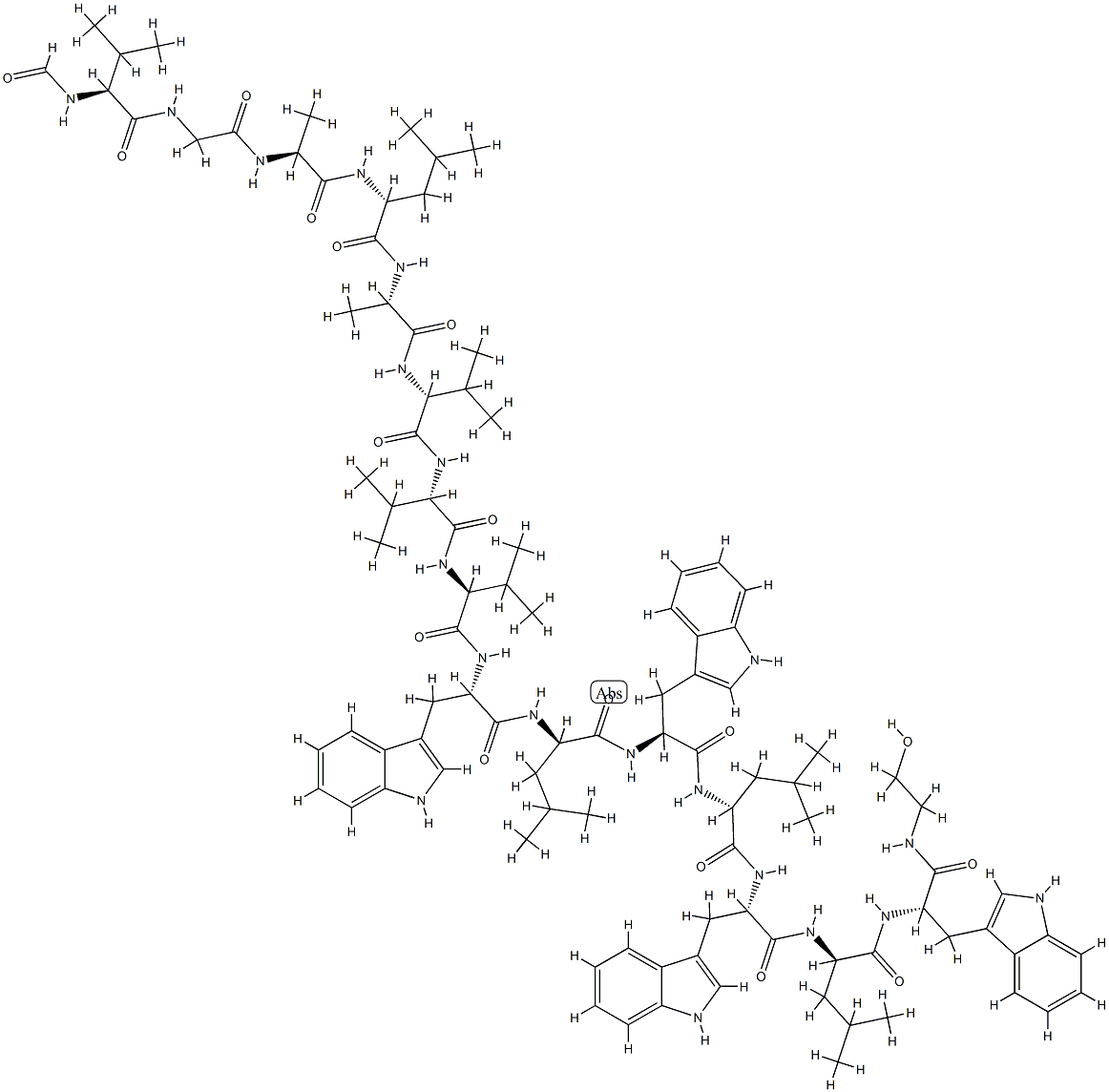
- Chemical Name:GRAMICIDIN
- CAS:1405-97-6
- MF:C99H140N20O17
- Structure:
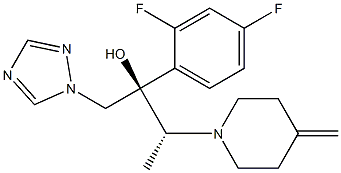
- Chemical Name:Efinaconazole
- CAS:164650-44-6
- MF:C18H22F2N4O
- Structure:
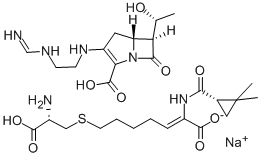
- Chemical Name:IMIPENEM
- CAS:85960-17-4
- MF:C16H25N2O5S.C12H18N4O4.Na
- Structure:
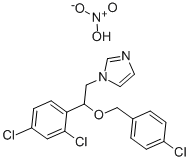
- Chemical Name:Econazole nitrate
- CAS:24169-02-6
- MF:C18H16Cl3N3O4
- Structure:
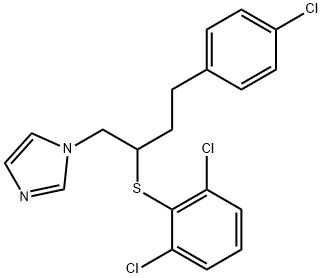
- Chemical Name:Butoconazole
- CAS:64872-76-0
- MF:C19H17Cl3N2S
- Structure:
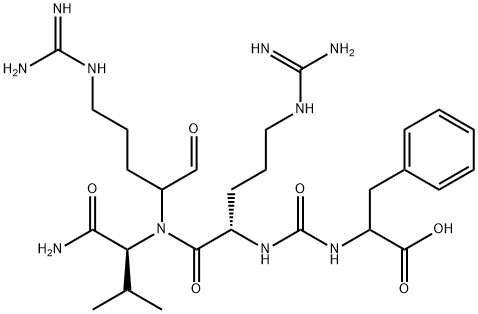
- Chemical Name:ANTIPAIN HYDROCHLORIDE DIHYDRATE
- CAS:37691-11-5
- MF:C27H44N10O6
- Structure:
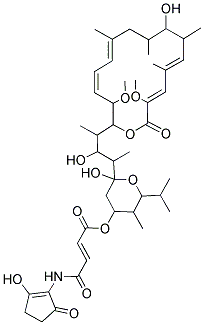
- Chemical Name:BAFILOMYCIN B1
- CAS:88899-56-3
- MF:C44H65NO13
- Structure:
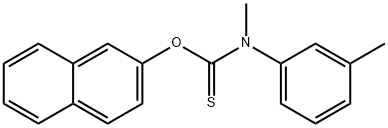
- Chemical Name:Tolnaftate
- CAS:2398-96-1
- MF:C19H17NOS
- Structure:
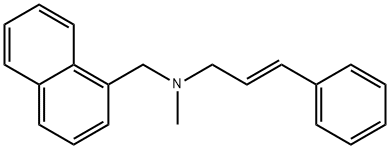
- Chemical Name:Naftifine
- CAS:65472-88-0
- MF:C21H21N
- Structure:
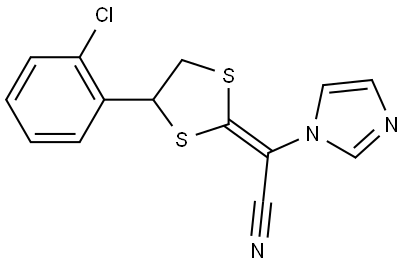
- Chemical Name:Lanoconazole
- CAS:101530-10-3
- MF:C14H10ClN3S2
- Structure:
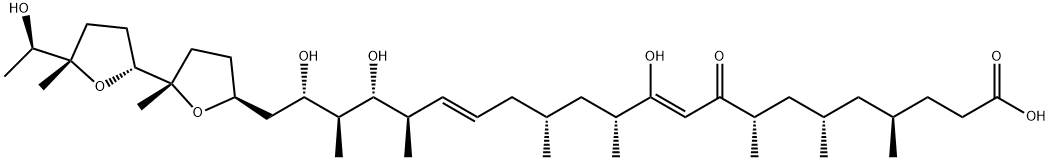
- Chemical Name:IONOMYCIN
- CAS:56092-81-0
- MF:C41H72O9
- Structure:
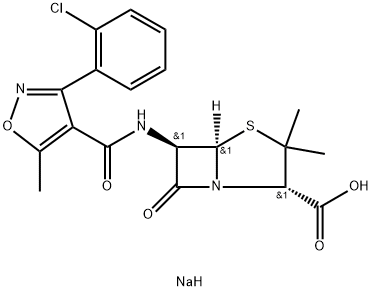
- Chemical Name:Cloxacillin-13C4 SodiuM Salt
- CAS:642-78-4
- MF:C19H19ClN3NaO5S
- Structure:
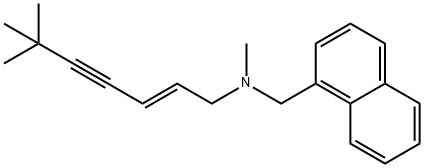
- Chemical Name:Terbinafine
- CAS:91161-71-6
- MF:C21H25N
- Structure:
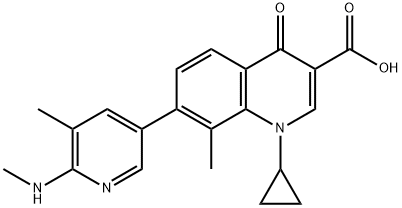
- Chemical Name:Ozenoxacin
- CAS:245765-41-7
- MF:C21H21N3O3
- Structure:
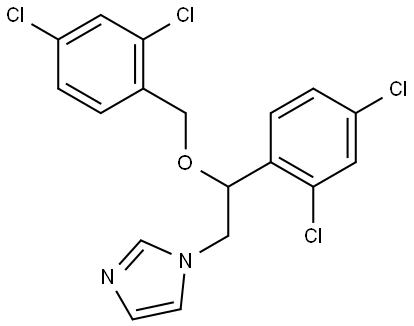
- Chemical Name:Miconazole
- CAS:22916-47-8
- MF:C18H14Cl4N2O
- Structure:
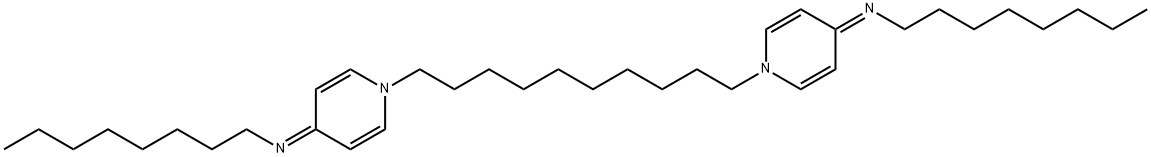
- Chemical Name:Octenidine
- CAS:71251-02-0
- MF:C36H62N4
- Structure:
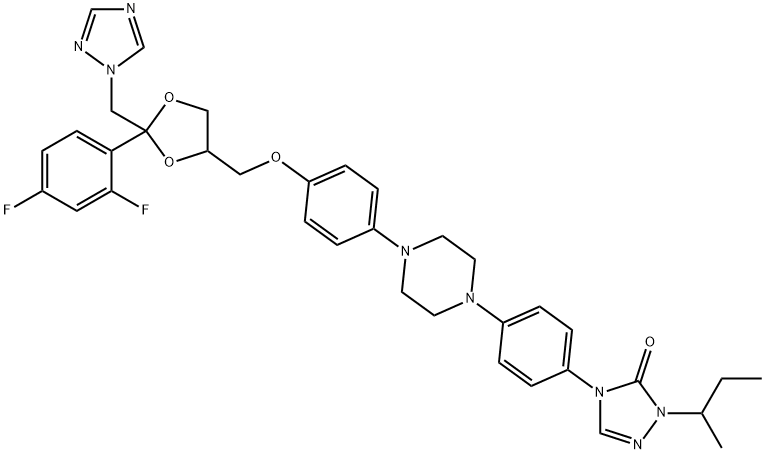
- Chemical Name:SAPERCONAZOLE
- CAS:110588-57-3
- MF:C35H38F2N8O4
- Structure:
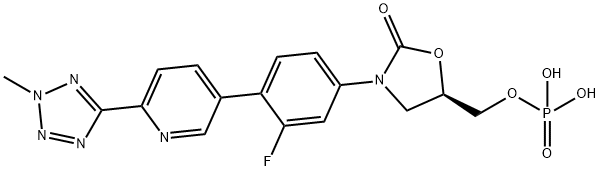
- Chemical Name:Tedizolid Phosphate
- CAS:856867-55-5
- MF:C17H16FN6O6P
- Structure:
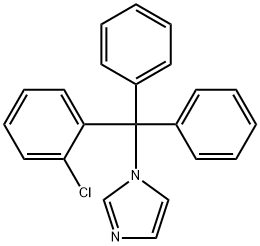
- Chemical Name:Clotrimazole
- CAS:23593-75-1
- MF:C22H17ClN2
- Structure:
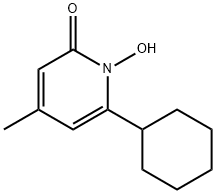
- Chemical Name:Ciclopirox
- CAS:29342-05-0
- MF:C12H17NO2
- Structure:
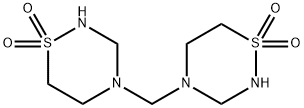
- Chemical Name:Taurolidine
- CAS:19388-87-5
- MF:C7H16N4O4S2
- Structure:
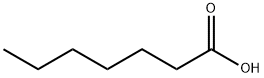
- Chemical Name:Heptanoic acid
- CAS:111-14-8
- MF:C7H14O2
- Structure:
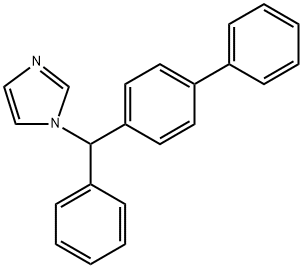
- Chemical Name:Bifonazole
- CAS:60628-96-8
- MF:C22H18N2
- Structure:
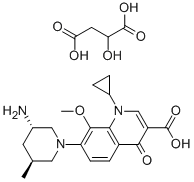
- Chemical Name:Butanedioicacid,2-hydroxy-,compd.with7-[(3S,5S)-3-amino-5-methyl-1-piperidinyl]-1-cyclopropyl-1,4-dihydro-8-methoxy-4-oxo-3-quinolinecarboxylicacid
- CAS:951163-60-3
- MF:C20H25N3O4.C4H6O5
- Structure:
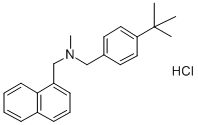
- Chemical Name:Butenafine hydrochloride
- CAS:101827-46-7
- MF:C23H27N.ClH
- Structure:
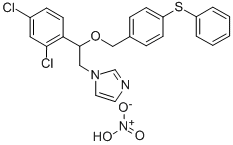
- Chemical Name:Fenticonazole nitrate
- CAS:73151-29-8
- MF:C24H21Cl2N3O4S
- Structure:
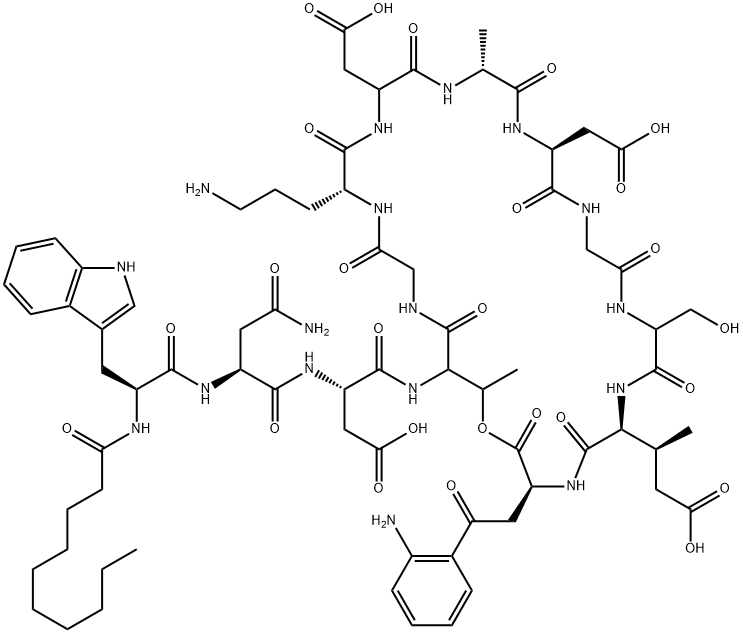
- Chemical Name:Daptomycin
- CAS:103060-53-3
- MF:C72H101N17O26
- Structure:
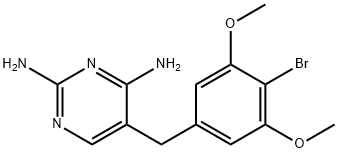
- Chemical Name:Brodimoprim
- CAS:56518-41-3
- MF:C13H15BrN4O2
- Structure:
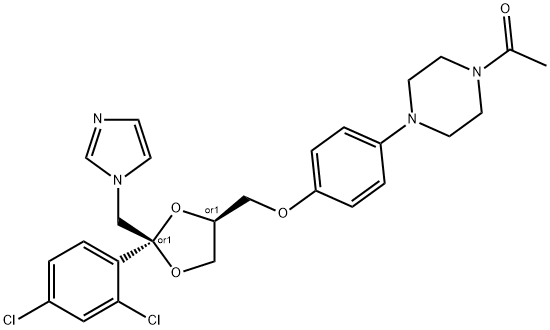
- Chemical Name:Ketoconazole
- CAS:65277-42-1
- MF:C26H28Cl2N4O4
- Chemical Name:PHLEOMYCIN
- CAS:11006-33-0
- MF:C55H86N20O21S2
- Structure:
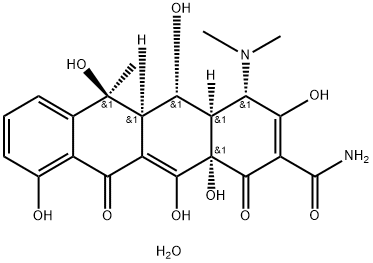
- Chemical Name:Oxytetracycline dihydrate
- CAS:6153-64-6
- MF:C22H26N2O10
- Structure:
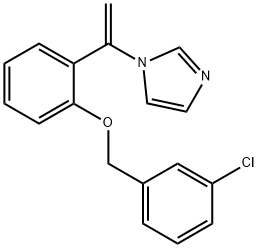
- Chemical Name:CROCONAZOLE
- CAS:77175-51-0
- MF:C18H15ClN2O
- Structure:
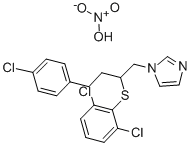
- Chemical Name:Butoconazole nitrate
- CAS:64872-77-1
- MF:C19H18Cl3N3O3S
- Structure:
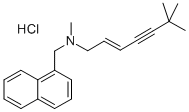
- Chemical Name:Terbinafine Hydrochloride
- CAS:78628-80-5
- MF:C21H26ClN
- Structure:
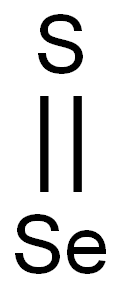
- Chemical Name:Selenium sulfide
- CAS:7446-34-6
- MF:SSe
- Structure:
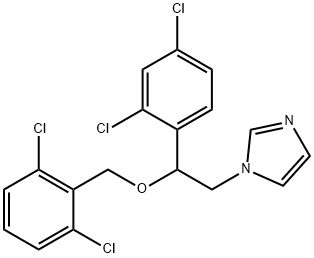
- Chemical Name:Isoconazole
- CAS:27523-40-6
- MF:C18H14Cl4N2O
- Structure:
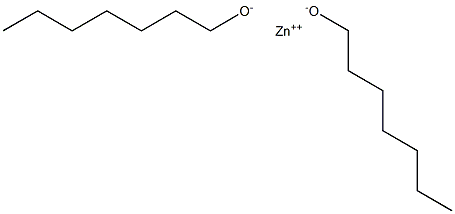
- Chemical Name:Zinc heptylate
- CAS:
- MF:C14H30O2Zn
- Structure:
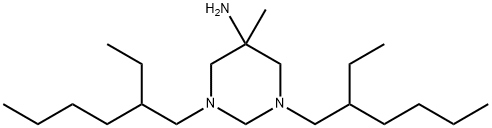
- Chemical Name:Hexetidine
- CAS:141-94-6
- MF:C21H45N3
- Structure:
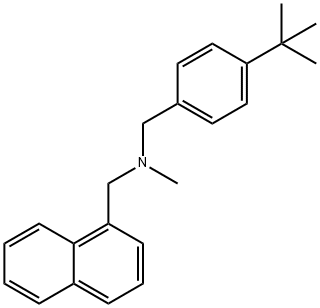
- Chemical Name:Butenafine
- CAS:101828-21-1
- MF:C23H27N
- Structure:
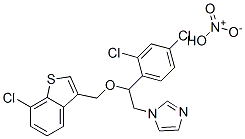
- Chemical Name:Sertaconazole nitrate
- CAS:99592-39-9
- MF:C20H15Cl3N2OS.HNO3
- Structure:
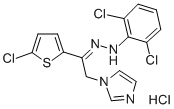
- Chemical Name:Zinoconazole hydrochloride
- CAS:80168-44-1
- MF:C15H11Cl3N4S.ClH
- Structure:
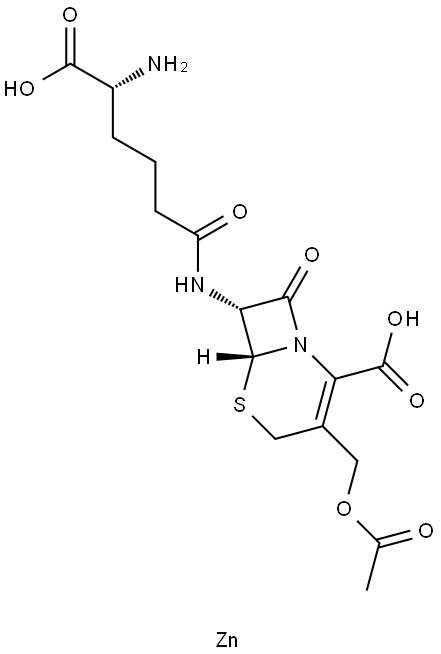
- Chemical Name:CEPHALOSPORIN C ZINC SALT
- CAS:59143-60-1
- MF:C16H21N3O8S.Zn
- Structure:
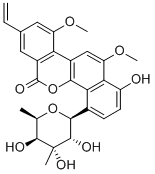
- Chemical Name:CHRYSOMYCIN A
- CAS:82196-88-1
- MF:C28H28O9
- Structure:
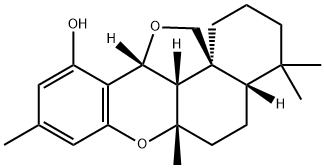
- Chemical Name:Siccanin
- CAS:22733-60-4
- MF:C22H30O3
- Structure:
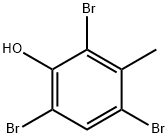
- Chemical Name:2,4,6-TRIBROMO-3-METHYLPHENOL
- CAS:4619-74-3
- MF:C7H5Br3O
- Structure:
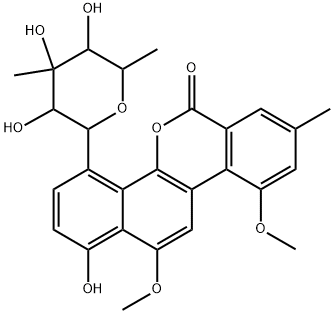
- Chemical Name:CHRYSOMYCIN B
- CAS:83852-56-6
- MF:C27H28O9
- Structure:
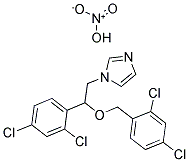
- Chemical Name:MICONAZOLE NITRATE
- CAS:75319-48-1
- MF:C18H15Cl4N3O4
- Structure:
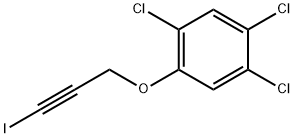
- Chemical Name:HALOPROGIN
- CAS:777-11-7
- MF:C9H4Cl3IO
- Structure:
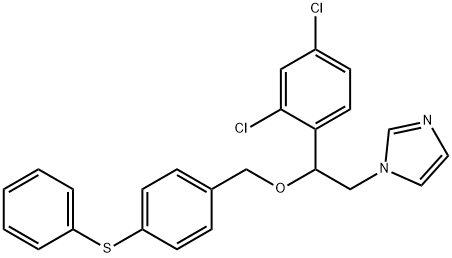
- Chemical Name:Fenticonazole
- CAS:72479-26-6
- MF:C24H20Cl2N2OS
- Structure:
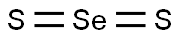
- Chemical Name:Selenium sulfide
- CAS:7488-56-4
- MF:S2Se
- Structure:
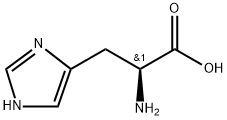
- Chemical Name:POLY-L-HISTIDINE
- CAS:61857-39-4
- MF:C6H9N3O2
- Structure:
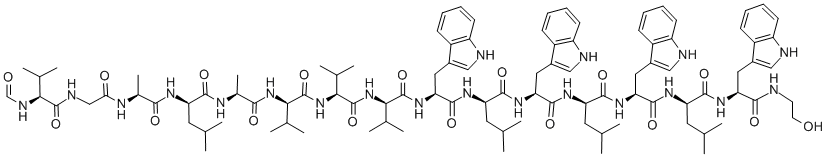
- Chemical Name:GRAMICIDIN A
- CAS:11029-61-1
- MF:C99H140N20O17
- Structure:
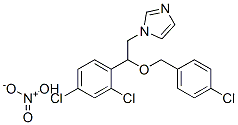
- Chemical Name:Econazole nitrate
- CAS:68797-31-9
- MF:C18H16Cl3N3O4
- Structure:
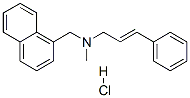
- Chemical Name:Naftifine hydrochloride
- CAS:65473-14-5
- MF:C21H22ClN
- Chemical Name:SULCONAZOLE NITRATE FPC(CRM STANDARD)
- CAS:
- MF:
- Structure:
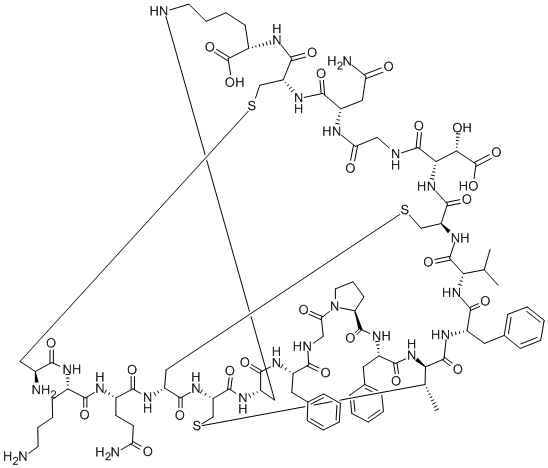
- Chemical Name:Lancovutide
- CAS:1391-36-2
- MF:C89H125N23O25S3
- Structure:
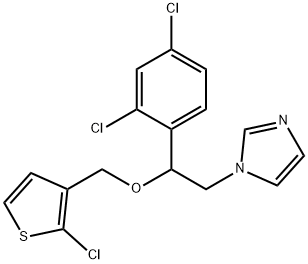
- Chemical Name:Vagistat
- CAS:65899-73-2
- MF:C16H13Cl3N2OS
- Structure:
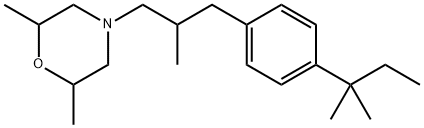
- Chemical Name:Amorolfine
- CAS:67467-83-8
- MF:C21H35NO
- Structure:

- Chemical Name:Selenium sulfide
- CAS:56093-45-9
- MF:H4SSe
- Structure:
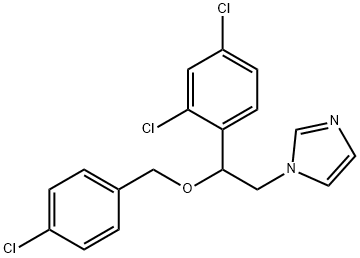
- Chemical Name:Econazole
- CAS:27220-47-9
- MF:C18H15Cl3N2O
- Structure:
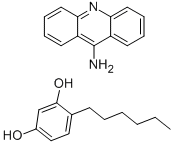
- Chemical Name:acrisorcin
- CAS:7527-91-5
- MF:C13H10N2.C12H18O2