Metal oxide
Metal oxides, especially metal oxides having semiconductor properties are the effective catalysts of oxidation - reduction reaction. Industrial catalysts typically contain one or more metal oxide component, called as a composite metal oxide catalyst. Generally speaking, there is at least one transition metal oxide contained it with each component forming mixture with each other at the molecular level and have interaction with each other, adjusting the electrical properties and surface acidity of the catalyst, improving the catalytic activity and selectivity.、
There are two situations regarding to the decomposition products of the metal oxide:
1. Decomposition into elemental metals and oxygen; the general principle of such reaction is that, more active the metal is , the more stable formed metal oxide will be, the more difficult formed metal oxide can be subject to decomposition; otherwise easily prone to be subject to decomposition. Only inert metal oxide can be subject to thermal decomposition; for example, mercuric oxide, silver oxide, etc; there is also some kinds of metal oxide can be subject to powered decomposition when being in the molten state.
2. High-valence metal oxide will be decomposed into low-valence oxide and oxygen when being heated metal oxide.
- Structure:
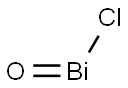
- Chemical Name:BISMUTH OXYCHLORIDE
- CAS:7787-59-9
- MF:BiClO
- Structure:

- Chemical Name:Rhodium oxide
- CAS:12036-35-0
- MF:H2ORh
- Structure:

- Chemical Name:BERYLLIUM OXIDE
- CAS:1304-56-9
- MF:BeO
- Structure:
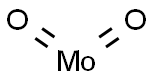
- Chemical Name:Molybdenum(IV) oxide
- CAS:18868-43-4
- MF:MoO2
- Structure:
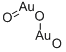
- Chemical Name:Digold trioxide
- CAS:1303-58-8
- MF:Au2O3
- Structure:

- Chemical Name:Aluminum oxide
- CAS:11092-32-3
- MF:AlO2
- Structure:
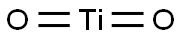
- Chemical Name:TITANIUM DIOXIDE
- CAS:1317-70-0
- MF:O2Ti
- Structure:
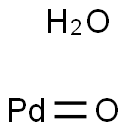
- Chemical Name:PALLADIUM(II) OXIDE
- CAS:64109-12-2
- MF:OPd
- Structure:
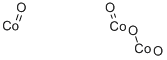
- Chemical Name:Tricobalt tetraoxide
- CAS:1308-06-1
- MF:Co3O4-2
- Structure:
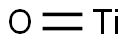
- Chemical Name:TITANIUM MONOXIDE
- CAS:12137-20-1
- MF:OTi
- Structure:
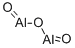
- Chemical Name:ALUMINUM OXIDE
- CAS:1302-74-5
- MF:Al2O3
- Structure:
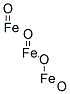
- Chemical Name:Iron oxide black
- CAS:1309-38-2
- MF:Fe3O4
- Structure:
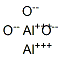
- Chemical Name:Aluminum oxide
- CAS:1344-28-1
- MF:Al2O3
- Structure:
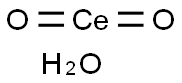
- Chemical Name:CERIUM(IV) OXIDE
- CAS:23322-64-7
- MF:CeH2O3
- Structure:
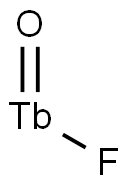
- Chemical Name:TERBIUM FLUORIDE
- CAS:21031-92-5
- MF:F3Tb
- Structure:
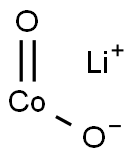
- Chemical Name:LITHIUM COBALT(III) OXIDE
- CAS:12190-79-3
- MF:CoLiO2
- Structure:
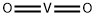
- Chemical Name:VANADIUM(IV) OXIDE
- CAS:12036-21-4
- MF:O2V
- Structure:
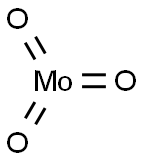
- Chemical Name:Molybdenum trioxide
- CAS:1313-27-5
- MF:MoO3
- Structure:
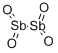
- Chemical Name:ANTIMONY (IV) OXIDE
- CAS:1332-81-6
- MF:O4Sb2
- Structure:

- Chemical Name:Lithium nickel manganese cobalt oxide
- CAS:346417-97-8
- MF:CoH3LiMnNiO
- Structure:
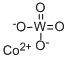
- Chemical Name:COBALT TUNGSTATE
- CAS:10101-58-3
- MF:CoO4W
- Chemical Name:High Purity Alpha Alumina(α)
- CAS:
- MF:
- Structure:
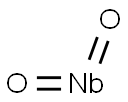
- Chemical Name:NIOBIUM (IV) OXIDE
- CAS:12034-59-2
- MF:NbO2
- Structure:
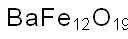
- Chemical Name:BARIUM FERRITE NANOPOWDER
- CAS:12047-11-9
- MF:BaFeH4O
- Structure:
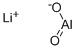
- Chemical Name:LITHIUM ALUMINATE
- CAS:12003-67-7
- MF:AlLiO2
- Structure:

- Chemical Name:Bismuth vanadate
- CAS:14059-33-7
- MF:BiO4V
- Chemical Name:Lanthanum cerium oxide
- CAS:
- MF:
- Structure:
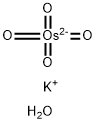
- Chemical Name:Potassium osmate(VI) dihydrate
- CAS:10022-66-9
- MF:H2KO5Os-
- Structure:
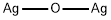
- Chemical Name:Silver oxide
- CAS:20667-12-3
- MF:Ag2O
- Structure:
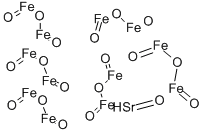
- Chemical Name:STRONTIUM FERRITE
- CAS:12023-91-5
- MF:Fe12O19Sr
- Structure:
- Chemical Name:COPPER TUNGSTATE
- CAS:13587-35-4
- MF:CuH2OW
- Structure:
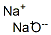
- Chemical Name:Sodium oxide
- CAS:1313-59-3
- MF:Na2O
- Structure:
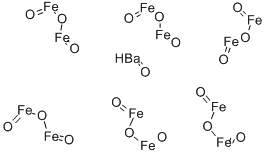
- Chemical Name:BARIUM FERRITE
- CAS:11138-11-7
- MF:BaO.6Fe2O3
- Structure:
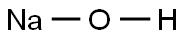
- Chemical Name:SODIUM DEUTEROXIDE
- CAS:14014-06-3
- MF:HNaO
- Chemical Name:Iron Oxide Black
- CAS:12227-89-3
- MF:Fe3O4
- Structure:
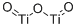
- Chemical Name:Titanium(III) oxide
- CAS:1344-54-3
- MF:O3Ti2
- Structure:
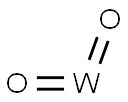
- Chemical Name:TUNGSTEN (IV) OXIDE
- CAS:12036-22-5
- MF:O2W
- Structure:
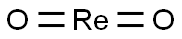
- Chemical Name:RHENIUM(IV) OXIDE
- CAS:12036-09-8
- MF:O2Re
- Chemical Name:Cobalt green
- CAS:
- MF:
- Structure:
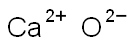
- Chemical Name:CALCIUM OXIDE
- CAS:73018-51-6
- MF:CaO
- Structure:
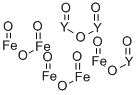
- Chemical Name:IRON YTTRIUM OXIDE
- CAS:12063-56-8
- MF:Fe5O12Y3
- Structure:
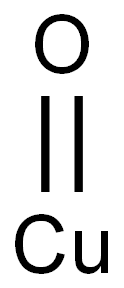
- Chemical Name:Cupric oxide
- CAS:1317-38-0
- MF:CuO
- Structure:
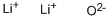
- Chemical Name:Lithium oxide
- CAS:12057-24-8
- MF:Li2O
- Structure:
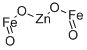
- Chemical Name:ZINC IRON OXIDE
- CAS:12063-19-3
- MF:Fe2O4Zn
- Structure:
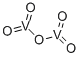
- Chemical Name:Vanadium(V) oxide
- CAS:1314-62-1
- MF:O5V2
- Structure:

- Chemical Name:TITANIUM OXIDE
- CAS:12065-65-5
- MF:H2OTi
- Structure:
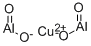
- Chemical Name:COPPER ALUMINUM OXIDE
- CAS:12042-92-1
- MF:Al2CuO4
- Structure:
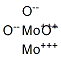
- Chemical Name:molybdenum sesquioxide
- CAS:1313-29-7
- MF:Mo2O3
- Structure:
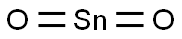
- Chemical Name:Stannic oxide
- CAS:18282-10-5
- MF:O2Sn
- Structure:
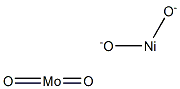
- Chemical Name:NICKEL MOLYBDATE
- CAS:14177-55-0
- MF:MoNiO4
- Structure:
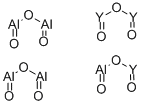
- Chemical Name:YTTRIUM ALUMINUM OXIDE
- CAS:12005-21-9
- MF:Al5O12Y3
- Structure:
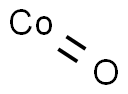
- Chemical Name:Cobalt oxide
- CAS:1307-96-6
- MF:CoO
- Structure:
- Chemical Name:CERIUM ZIRCONIUM OXIDE
- CAS:53169-24-7
- MF:CeH2OZr
- Structure:

- Chemical Name:AMMONIUM TUNGSTATE
- CAS:11140-77-5
- MF:H6NOW+
- Chemical Name:Iron Oxide Green
- CAS:222961-35-5
- MF:
- Structure:
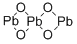
- Chemical Name:lead oxide
- CAS:1314-41-6
- MF:O4Pb3
- Structure:
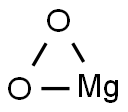
- Chemical Name:Magnesium dioxide
- CAS:14452-57-4
- MF:MgO2
- Structure:

- Chemical Name:ALUMINUM ZIRCONATE
- CAS:70692-95-4
- MF:Al2O9Zr3
- Structure:
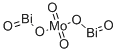
- Chemical Name:BISMUTH MOLYBDATE
- CAS:13565-96-3
- MF:Bi2MoO6
- Structure:

- Chemical Name:THALLIUM (I) OXIDE
- CAS:1314-12-1
- MF:OTl2
- Structure:
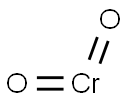
- Chemical Name:chromium dioxide
- CAS:12018-01-8
- MF:CrO2
- Structure:
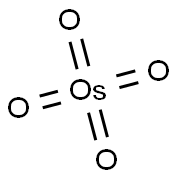
- Chemical Name:Osmium tetroxide
- CAS:20816-12-0
- MF:O4Os
- Structure:

- Chemical Name:Holmium oxide
- CAS:39455-61-3
- MF:H2HoO
- Structure:
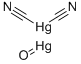
- Chemical Name:MERCURIC OXYCYANIDE
- CAS:1335-31-5
- MF:C2Hg2N2O
- Structure:
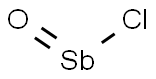
- Chemical Name:ANTIMONY OXYCHLORIDE
- CAS:7791-08-4
- MF:ClOSb
- Structure:
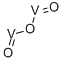
- Chemical Name:VANADIUM (III) OXIDE
- CAS:1314-34-7
- MF:O3V2
- Structure:
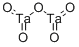
- Chemical Name:Tantalum pentoxide
- CAS:1314-61-0
- MF:O5Ta2
- Structure:
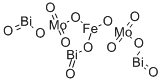
- Chemical Name:BISMUTH IRON MOLYBDENUM OXIDE
- CAS:59393-06-5
- MF:Bi3FeMo2O12
- Structure:
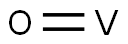
- Chemical Name:VANADIUM OXIDE
- CAS:12035-98-2
- MF:OV
- Structure:
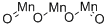
- Chemical Name:MANGANESE OXIDE
- CAS:1309-55-3
- MF:Mn3O4
- Structure:
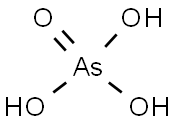
- Chemical Name:ARSENIC(V) OXIDE HYDRATE
- CAS:12044-50-7
- MF:As2H2O6
- Structure:
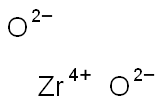
- Chemical Name:Zirconium dioxide
- CAS:1314-23-4
- MF:O2Zr
- Structure:
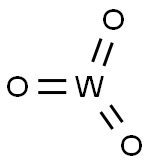
- Chemical Name:Tungsten trioxide
- CAS:1314-35-8
- MF:O3W
- Structure:
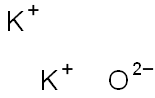
- Chemical Name:Potassium oxide
- CAS:12136-45-7
- MF:K2O
- Structure:
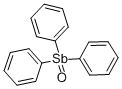
- Chemical Name:TRIPHENYLANTIMONY OXIDE
- CAS:4756-75-6
- MF:C18H15OSb
- Structure:
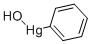
- Chemical Name:HYDROXYPHENYLMERCURY
- CAS:100-57-2
- MF:C6H6HgO
- Structure:
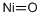
- Chemical Name:Nickel(II) oxide
- CAS:1313-99-1
- MF:NiO
- Structure:
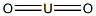
- Chemical Name:URANIUM(IV) OXIDE
- CAS:1344-57-6
- MF:O2U
- Structure:
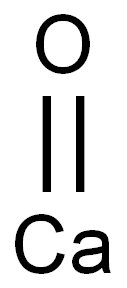
- Chemical Name:Calcium oxide
- CAS:1305-78-8
- MF:CaO
- Structure:
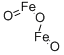
- Chemical Name:Ferric oxide
- CAS:1309-37-1
- MF:Fe2O3
- Structure:

- Chemical Name:Iron oxide
- CAS:1332-37-2
- MF:Fe2O3
- Structure:
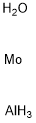
- Chemical Name:ALUMINUM MOLYBDATE
- CAS:15123-80-5
- MF:AlH5MoO
- Structure:
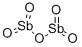
- Chemical Name:Diantimony pentoxide
- CAS:1314-60-9
- MF:O5Sb2
- Structure:
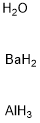
- Chemical Name:BARIUM ALUMINATE
- CAS:12004-04-5
- MF:AlBaH7O
- Structure:
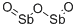
- Chemical Name:Diantimony trioxide
- CAS:1309-64-4
- MF:O3Sb2
- Structure:
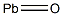
- Chemical Name:Lead monoxide
- CAS:1317-36-8
- MF:OPb
- Structure:
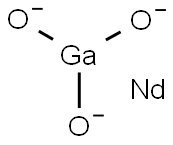
- Chemical Name:NEODYMIUM GALLIUM OXIDE
- CAS:12207-22-6
- MF:GaNdO3-3
- Structure:
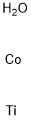
- Chemical Name:COBALT TITANATE
- CAS:12017-01-5
- MF:CoH2OTi
- Structure:
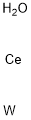
- Chemical Name:CERIUM TUNGSTATE
- CAS:13454-74-5
- MF:CeH2OW
- Structure:
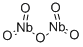
- Chemical Name:Niobium oxide
- CAS:1313-96-8
- MF:Nb2O5
- Structure:
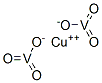
- Chemical Name:COPPER VANADATE
- CAS:14958-34-0
- MF:CuO6V2
- Structure:
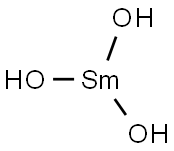
- Chemical Name:SAMARIUM(III) HYDROXIDE HYDRATE
- CAS:20403-06-9
- MF:H5O4Sm
- Structure:
- Chemical Name:BARIUM NIOBIUM OXIDE
- CAS:12009-14-2
- MF:BaH4NbO
- Structure:
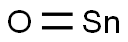
- Chemical Name:TIN(II) OXIDE
- CAS:21651-19-4
- MF:OSn
- Structure:
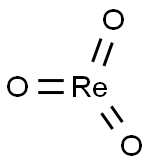
- Chemical Name:RHENIUM (VI) OXIDE
- CAS:1314-28-9
- MF:O3Re
- Structure:
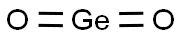
- Chemical Name:Germanium oxide
- CAS:1310-53-8
- MF:GeO2
- Structure:
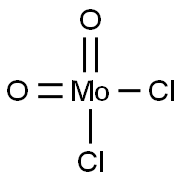
- Chemical Name:MOLYBDENUM(VI) DICHLORIDE DIOXIDE
- CAS:13637-68-8
- MF:Cl2MoO2
- Structure:
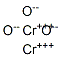
- Chemical Name:Chromium(III) oxide
- CAS:1308-38-9
- MF:Cr2O3
- Structure:
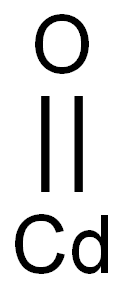
- Chemical Name:Cadmium oxide
- CAS:1306-19-0
- MF:CdO
- Structure:
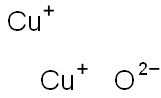
- Chemical Name:Cuprous oxide
- CAS:1317-39-1
- MF:Cu2O