tert-Butyl chloride is a colourless liquid. Miscible with alcohol and ether, insoluble in water. It is flammable and releases toxic phosgene when heated. Reacts violently with oxidizing agents. More toxic than 1-chlorobutane. tert-Butyl Chloride has a wide range of applications in the fields of medicine, pesticides, rubber, plastic additives and other chemical industries.
2-Chloro-2-methylpropane is an alkylating agent that is used to functionalize molecules with tert-butyl group.
It serves as an effective chlorinating agent, in combination with the ionic liquid, 1-butyl-3-methylimidazolium bromide for converting alcohols into chlorides.
It can also be used to prepare tert-butyl ethers by treating with corresponding alcohols.
tert-Butyl chloride plays an important role as a starting material to perform nucleophilic substitution reactions in order to prepare alcohol and alkoxides salts. It is used as an alkylating agent for the introduction of tert-butyl group and is also involved in Friedel-Crafts reactions. It is employed as an intermediate for the synthesis of agrochemicals and pharmaceuticals.
Flammability and Explosibility
Flammable
Questionable
carcinogen with experimental neoplastigenic
data. Dangerous fire hazard when exposed
to heat, flame (sparks), and oxidlzers. To
fight fue, use water, spray, fog, alcohol
foam, dry chemical. When heated to
decomposition it emits toxic fumes of Cl-.
See also CHLORINATED
HYDROCARBONS, ALIPHATIC.
tert-Butyl chloride is prepared from tertbutyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction.
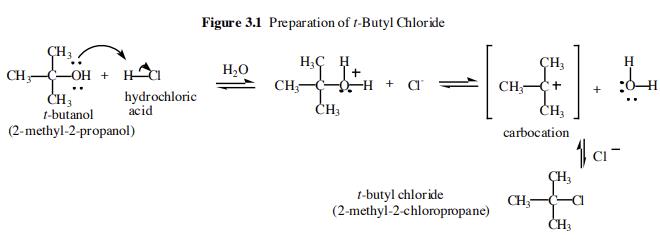
The first step of the overall reaction is an acid-base reaction between the t-butanol and the hydrochloric acid. The t-butanol is a weak base and the hydrochloric acid is a strong acid. The alcoholic oxygen becomes fully protonated and so the equilibrium lies far to the right. In the second step we have the slow loss of water to form a carbocation intermediate. This species is very reactive and is immediately attacked by the chloride ion liberated in the first step to form tert-Butyl chloride.
Purification methods commonly used for other alkyl halides lead to decomposition. Some impurities can be removed by photochlorination with a small amount of chlorine prior to use. The liquid is washed with ice water, dried with CaCl2 or CaCl2 + CaO and fractionally distilled. It has been further purified by repeated fractional crystallisation by partial freezing. [Beilstein 1 IV 288.]