Inorganic acid Esters
Inorganic acid ester is obtained through the reaction between alcohol and oxygen-containing organic acids or inorganic acid to generate esters (inorganic acid ester or organic acid esters) and water reaction. For example, sulfuric acid and ethanol, nitric acid and glycerol or pentaerythritol esters can have esterification reaction to generate important inorganic acids ester.
The compound obtained through the dehydration reaction between acid (inorganic and organic acids) and the alcohol is ester. It can be divided into inorganic acid esters (e.g., dimethyl sulfate, methyl hydrogen sulfate, glyceryltrinitrate, tributyl phosphate, etc.), and organic acid esters (e.g., ethyl acetate, vinyl acetate, and [alpha] methyl methacrylate ester, etc.) two categories. But the latter one is more important. In accordance with the intramolecular or intermolecular esterification of hydroxy acid (alcohol acid), it can also be divided into two kinds, lactone and lactide. Ester is generally neutral substance and can be subject to hydrolysis. Carboxylic acid ester can participate in a series of reactions including transesterification (alcoholysis), ammonolysis, reduction (hydrogenolysis) and the Grignard reaction.
Usually refers to a carboxylic acid ester with the general formula being RCOOR ', being the product with the hydrogen in the carboxyl group of carboxylic acid being replaced by hydrocarbon. The names of the esters are based on the names of corresponding carboxylic acid and alcohol or phenol such as "a certain acid ester." The cyclic ester is called lactone.
The chemical properties of ester are similar to acyl halide and acid anhydride. It is prone to have hydrolysis, alcoholysis and aminolysis reaction. The lower esters are aromatic volatile colorless liquid with the high-grade ester being solid. Esters are important solvents and synthetic chemicals with some kinds of ester itself being medicines. Depending on the type of acid, esters can be divided into inorganic and organic esters, such as hydrogen methyl sulfate (CH3OSO3H) for the former one and ethyl acetate CH3COOCH2CH3 for the later one; depending on the type of hydrocarbon, the ester can also be divided into fatty esters and aromatic esters and ring esters; ethyl acetate belongs to fatty esters; the phenyl acetate belongs to aromatic esters; while methyl furoate belongs to cyclic ester.
From the broad definition, the reaction between alcohols and acids for generating ester should be called as esterification. But usually it only refers to acid esterification and the esterification of alcohol is called as acylation. Esterification is reversible with the ester having hydrolysis with water to generate the original alcohol (or phenol) and acid. In general, the esterfication should be conducted in the presence of catalyst (hydrogen ion) and heating conditions, in order to shorten the time required for reaching equilibrium.
- Structure:
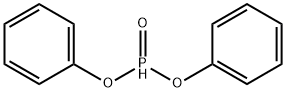
- Chemical Name:DIPHENYL PHOSPHITE
- CAS:4712-55-4
- MF:C12H11O3P
- Structure:
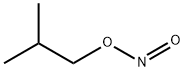
- Chemical Name:Isobutyl nitrite
- CAS:542-56-3
- MF:C4H9NO2
- Structure:
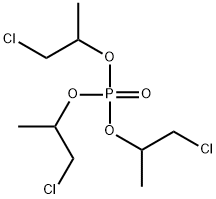
- Chemical Name:Tris(1-Chloro-2-Propyl) Phosphate
- CAS:13674-84-5
- MF:C9H18Cl3O4P
- Structure:
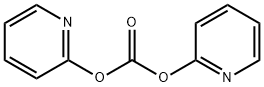
- Chemical Name:CARBONIC ACID DI-2-PYRIDYL ESTER
- CAS:1659-31-0
- MF:C11H8N2O3
- Structure:
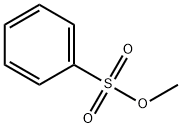
- Chemical Name:Methyl benzenesulfonate
- CAS:80-18-2
- MF:C7H8O3S
- Structure:
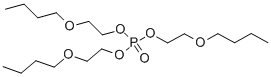
- Chemical Name:Tris(2-butoxyethyl) phosphate
- CAS:78-51-3
- MF:C18H39O7P
- Structure:
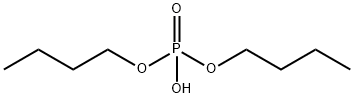
- Chemical Name:Dibutyl phosphate
- CAS:107-66-4
- MF:C8H19O4P
- Structure:
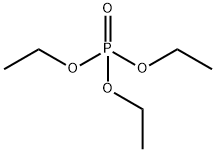
- Chemical Name:Triethyl phosphate
- CAS:78-40-0
- MF:C6H15O4P
- Structure:
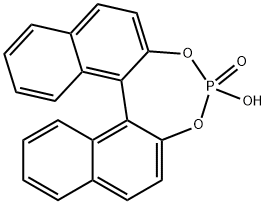
- Chemical Name:1,1'-Binaphthyl-2,2'-diyl hydrogenphosphate
- CAS:35193-63-6
- MF:C20H13O4P
- Structure:
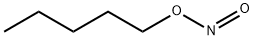
- Chemical Name:Pentyl nitrite
- CAS:463-04-7
- MF:C5H11NO2
- Structure:
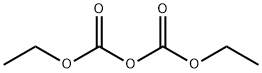
- Chemical Name:Diethyl pyrocarbonate
- CAS:1609-47-8
- MF:C6H10O5
- Structure:
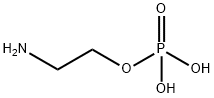
- Chemical Name:O-PHOSPHORYLETHANOLAMINE
- CAS:1071-23-4
- MF:C2H8NO4P
- Structure:
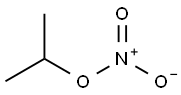
- Chemical Name:Isopropyl nitrate
- CAS:1712-64-7
- MF:C3H7NO3
- Structure:
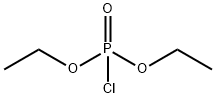
- Chemical Name:Diethyl chlorophosphate
- CAS:814-49-3
- MF:C4H10ClO3P
- Structure:
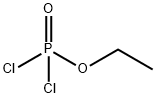
- Chemical Name:Ethyl dichlorophosphate
- CAS:1498-51-7
- MF:C2H5Cl2O2P
- Structure:
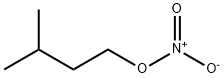
- Chemical Name:ISOAMYL NITRATE
- CAS:543-87-3
- MF:C5H11NO3
- Structure:
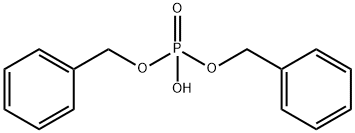
- Chemical Name:Dibenzyl phosphate
- CAS:1623-08-1
- MF:C14H15O4P
- Structure:
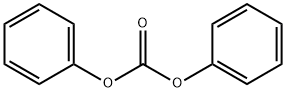
- Chemical Name:Diphenyl carbonate
- CAS:102-09-0
- MF:C13H10O3
- Structure:
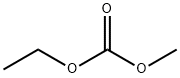
- Chemical Name:Ethyl methyl carbonate
- CAS:623-53-0
- MF:C4H8O3
- Structure:
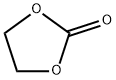
- Chemical Name:Ethylene carbonate
- CAS:96-49-1
- MF:C3H4O3
- Structure:
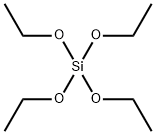
- Chemical Name:Ethyl silicate
- CAS:78-10-4
- MF:C8H20O4Si
- Structure:
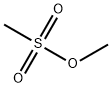
- Chemical Name:Methyl methanesulfonate
- CAS:66-27-3
- MF:C2H6O3S
- Structure:
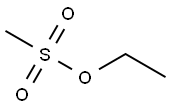
- Chemical Name:Ethyl methanesulfonate
- CAS:62-50-0
- MF:C3H8O3S
- Structure:
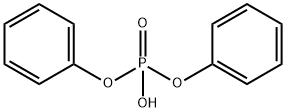
- Chemical Name:Diphenyl phosphate
- CAS:838-85-7
- MF:C12H11O4P
- Structure:
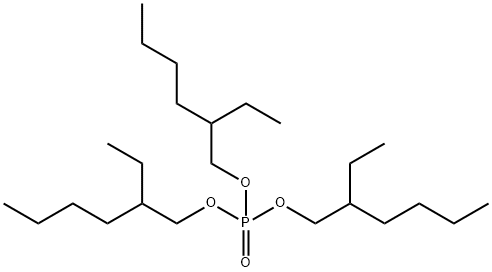
- Chemical Name: Tris(2-ethylhexyl) phosphate
- CAS:78-42-2
- MF:C24H51O4P
- Structure:
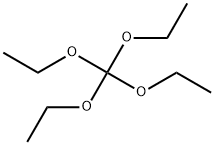
- Chemical Name:Tetraethyl orthocarbonate
- CAS:78-09-1
- MF:C9H20O4
- Structure:
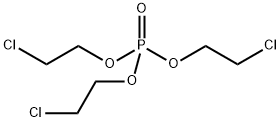
- Chemical Name:Tris(2-chloroethyl) phosphate
- CAS:115-96-8
- MF:C6H12Cl3O4P
- Structure:
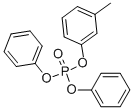
- Chemical Name:Cresyl diphenyl phosphate
- CAS:26444-49-5
- MF:C19H17O4P
- Structure:
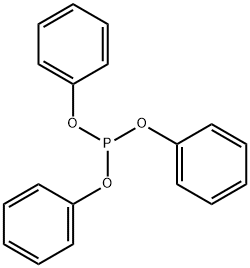
- Chemical Name:Triphenyl phosphite
- CAS:101-02-0
- MF:C18H15O3P
- Structure:
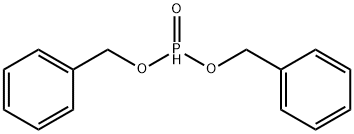
- Chemical Name:Dibenzyl phosphite
- CAS:17176-77-1
- MF:C14H15O3P
- Structure:
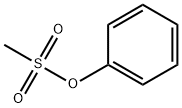
- Chemical Name:Phenyl methanesulfonate
- CAS:16156-59-5
- MF:C7H8O3S
- Structure:
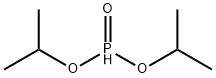
- Chemical Name:Diisopropyl phosphite
- CAS:1809-20-7
- MF:C6H15O3P
- Structure:
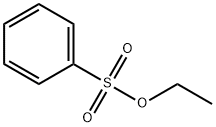
- Chemical Name:Ethyl benzenesulphonate
- CAS:515-46-8
- MF:C8H10O3S
- Structure:
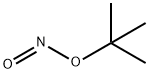
- Chemical Name:tert-Butyl nitrite
- CAS:540-80-7
- MF:C4H9NO2
- Structure:
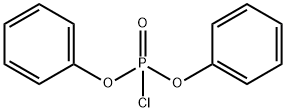
- Chemical Name:Diphenyl chlorophosphate
- CAS:2524-64-3
- MF:C12H10ClO3P
- Structure:
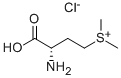
- Chemical Name:DL-METHIONINE METHYLSULFONIUM CHLORIDE
- CAS:1115-84-0
- MF:C6H14ClNO2S
- Structure:
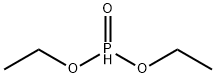
- Chemical Name:Diethyl phosphite
- CAS:762-04-9
- MF:C4H11O3P
- Structure:
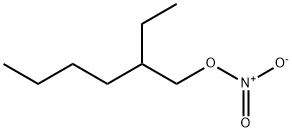
- Chemical Name:2-Ethylhexyl nitrate
- CAS:27247-96-7
- MF:C8H17NO3
- Structure:
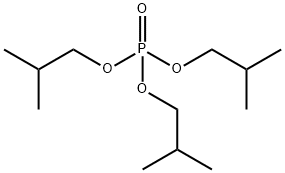
- Chemical Name:Triisobutyl phosphate
- CAS:126-71-6
- MF:C12H27O4P
- Structure:
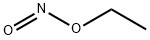
- Chemical Name:Ethyl nitrite
- CAS:109-95-5
- MF:C2H5NO2
- Structure:
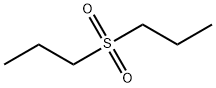
- Chemical Name:DIPROPYL SULFATE
- CAS:598-03-8
- MF:C6H14O2S
- Structure:
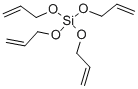
- Chemical Name:TETRAALLYLOXYSILANE
- CAS:1067-43-2
- MF:C12H20O4Si
- Structure:
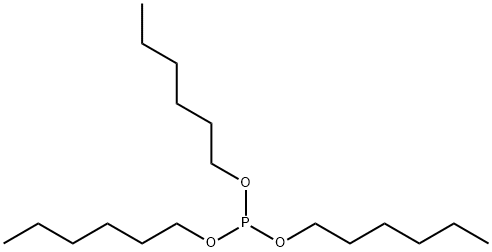
- Chemical Name:TRIHEXYL PHOSPHITE
- CAS:6095-42-7
- MF:C18H39O3P
- Structure:
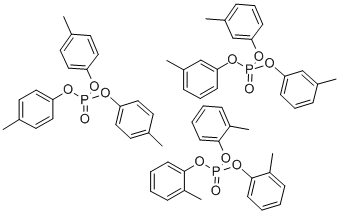
- Chemical Name:Tricresyl Phosphate
- CAS:1330-78-5
- MF:C21H21O4P
- Structure:
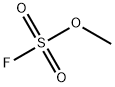
- Chemical Name:METHYL FLUOROSULFONATE
- CAS:421-20-5
- MF:CH3FO3S
- Structure:
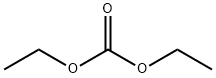
- Chemical Name:Diethyl carbonate
- CAS:105-58-8
- MF:C5H10O3
- Structure:
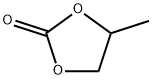
- Chemical Name:Propylene carbonate
- CAS:108-32-7
- MF:C4H6O3
- Structure:
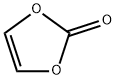
- Chemical Name:Vinylene carbonate
- CAS:872-36-6
- MF:C3H2O3
- Structure:
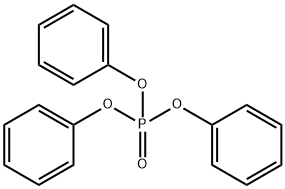
- Chemical Name:Triphenyl phosphate
- CAS:115-86-6
- MF:C18H15O4P
- Structure:
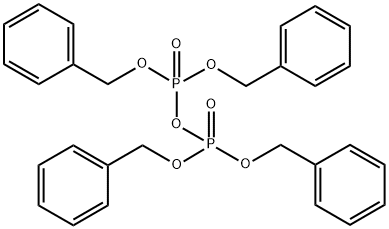
- Chemical Name:Tetrabenzyl pyrophosphate
- CAS:990-91-0
- MF:C28H28O7P2
- Structure:
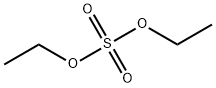
- Chemical Name:Diethyl sulfate
- CAS:64-67-5
- MF:C4H10O4S
- Structure:
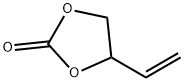
- Chemical Name:4-Vinyl-1,3-dioxolan-2-one
- CAS:4427-96-7
- MF:C5H6O3
- Structure:
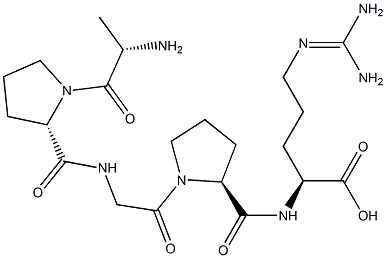
- Chemical Name:Alkaline Phosphatase
- CAS:9001-78-9
- MF:C21H36N8O6
- Structure:
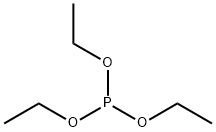
- Chemical Name:Triethyl phosphite
- CAS:122-52-1
- MF:C6H15O3P
- Structure:
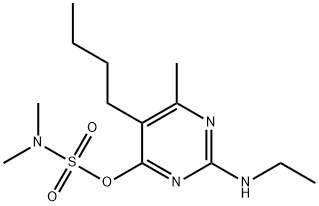
- Chemical Name:BUPIRIMATE
- CAS:41483-43-6
- MF:C13H24N4O3S
- Structure:
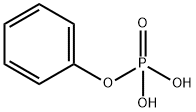
- Chemical Name:PHENYLPHOSPHORIC ACID
- CAS:701-64-4
- MF:C6H7O4P
- Structure:
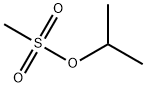
- Chemical Name:ISOPROPYL METHANESULFONATE
- CAS:926-06-7
- MF:C4H10O3S
- Structure:
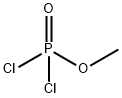
- Chemical Name:METHYL PHOSPHORODICHLORIDATE
- CAS:677-24-7
- MF:CH3Cl2O2P
- Structure:
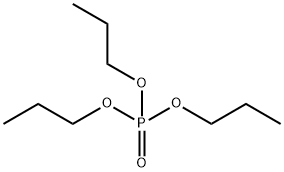
- Chemical Name:TRIPROPYL PHOSPHATE
- CAS:513-08-6
- MF:C9H21O4P
- Structure:
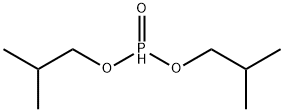
- Chemical Name:DIISOBUTYL PHOSPHITE
- CAS:1189-24-8
- MF:C8H19O3P
- Structure:
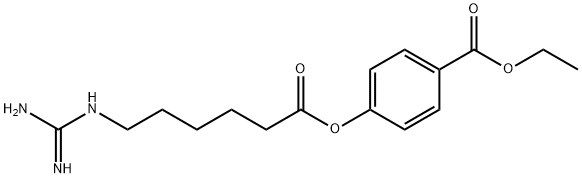
- Chemical Name:GABEXATE
- CAS:39492-01-8
- MF:C16H23N3O4
- Structure:
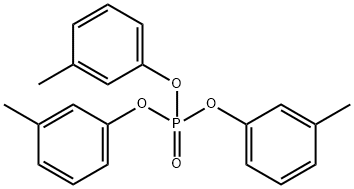
- Chemical Name:TRI-M-TOLYL PHOSPHATE
- CAS:563-04-2
- MF:C21H21O4P
- Structure:
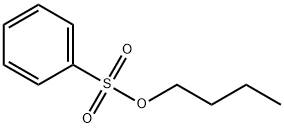
- Chemical Name:Butyl Benzenesulfonate
- CAS:80-44-4
- MF:C10H14O3S
- Structure:
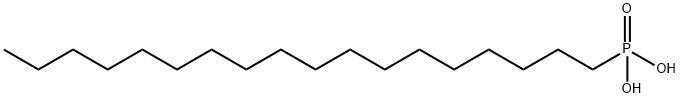
- Chemical Name:Octadecylphosphonic acid
- CAS:4724-47-4
- MF:C18H39O3P
- Structure:
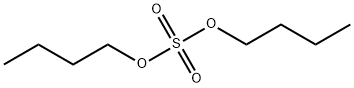
- Chemical Name:DI-N-BUTYL SULFATE
- CAS:625-22-9
- MF:C8H18O4S
- Structure:
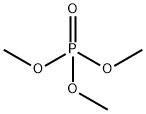
- Chemical Name:Trimethyl phosphate
- CAS:512-56-1
- MF:C3H9O4P
- Structure:

- Chemical Name:tert-Butylphenyl diphenyl phosphate
- CAS:56803-37-3
- MF:C22H23O4P
- Structure:
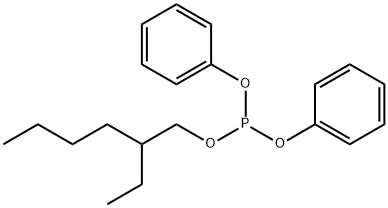
- Chemical Name:2-ETHYLHEXYL DIPHENYL PHOSPHITE
- CAS:15647-08-2
- MF:C20H27O3P
- Structure:
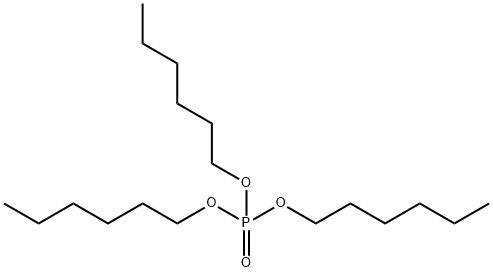
- Chemical Name:TRIHEXYL PHOSPHATE
- CAS:2528-39-4
- MF:C18H39O4P
- Structure:
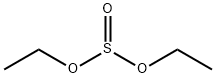
- Chemical Name:Diethyl sulfite
- CAS:623-81-4
- MF:C4H10O3S
- Structure:
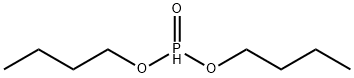
- Chemical Name:Dibutyl phosphite
- CAS:1809-19-4
- MF:C8H19O3P
- Structure:
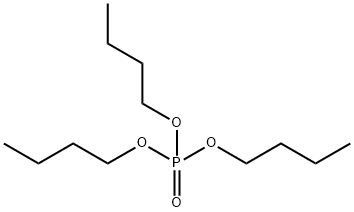
- Chemical Name:Tributyl phosphate
- CAS:126-73-8
- MF:C12H27O4P
- Structure:
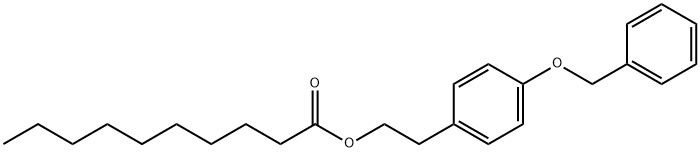
- Chemical Name:Decanoic acid, 2-[4-(phenylMethoxy)phenyl]ethyl ester
- CAS:848484-93-5
- MF:C25H34O3
- Structure:

- Chemical Name:Trixylyl phosphate
- CAS:25155-23-1
- MF:C8H11O4P
- Structure:
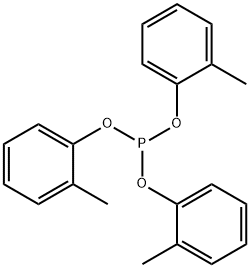
- Chemical Name:PHOSPHOROUS ACID TRI-O-CRESYL ESTER
- CAS:2622-08-4
- MF:C21H21O3P
- Structure:
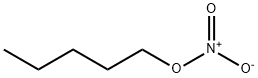
- Chemical Name:N-AMYL NITRATE
- CAS:1002-16-0
- MF:C5H11NO3
- Structure:
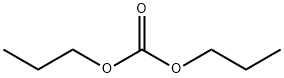
- Chemical Name:Dipropyl carbonate
- CAS:623-96-1
- MF:C7H14O3
- Structure:
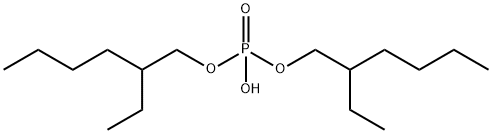
- Chemical Name:Bis(2-ethylhexyl) phosphate
- CAS:298-07-7
- MF:C16H35O4P
- Structure:
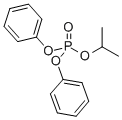
- Chemical Name:Diphenyl isopropylphenyl phosphate
- CAS:28108-99-8
- MF:C21H21O4P
- Structure:
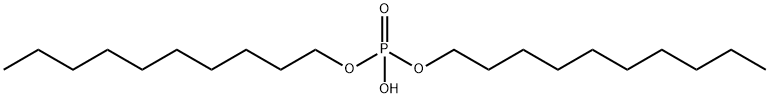
- Chemical Name:PHOSPHORIC ACID DI-N-DECYL ESTER
- CAS:7795-87-1
- MF:C20H43O4P
- Structure:
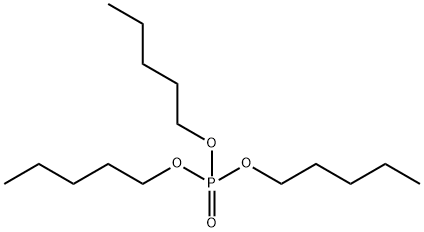
- Chemical Name:PHOSPHORIC ACID TRI-N-AMYL ESTER
- CAS:2528-38-3
- MF:C15H33O4P
- Structure:
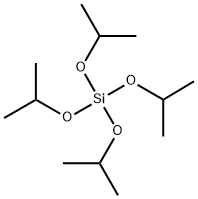
- Chemical Name:TETRAISOPROPYL ORTHOSILICATE
- CAS:1992-48-9
- MF:C12H28O4Si
- Structure:
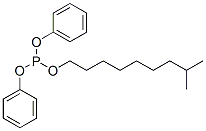
- Chemical Name:Isodecyl diphenyl phosphite
- CAS:26544-23-0
- MF:C22H31O3P
- Structure:
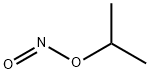
- Chemical Name:ISOPROPYL NITRITE
- CAS:541-42-4
- MF:C3H7NO2
- Structure:
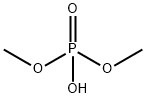
- Chemical Name:DIMETHYL PHOSPHATE
- CAS:813-78-5
- MF:C2H7O4P
- Structure:
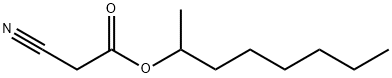
- Chemical Name: 2-Octyl cyanoacetate
- CAS:52688-08-1
- MF:C11H19NO2
- Structure:
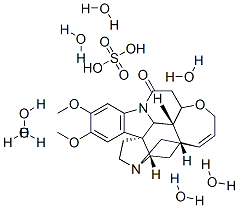
- Chemical Name:BRUCINE SULFATE HEPTAHYDRATE
- CAS:5787-00-8
- MF:C46H68N4O19S
- Structure:
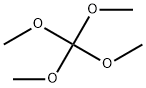
- Chemical Name:Tetramethoxymethane
- CAS:1850-14-2
- MF:C5H12O4
- Structure:
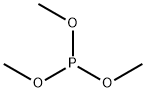
- Chemical Name:Trimethyl phosphite
- CAS:121-45-9
- MF:C3H9O3P
- Structure:
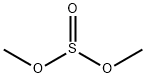
- Chemical Name:Dimethyl sulfite
- CAS:616-42-2
- MF:C2H6O3S
- Structure:
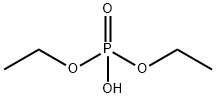
- Chemical Name:DIETHYL PHOSPHATE
- CAS:598-02-7
- MF:C4H11O4P
- Structure:
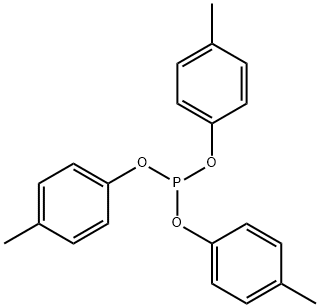
- Chemical Name:PHOSPHOROUS ACID TRIS(4-METHYLPHENYL) ESTER
- CAS:620-42-8
- MF:C21H21O3P
- Structure:
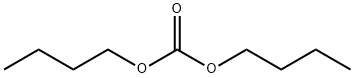
- Chemical Name:DIBUTYL CARBONATE
- CAS:542-52-9
- MF:C9H18O3
- Structure:
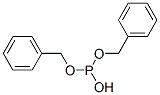
- Chemical Name:DIBENZYL PHOSPHITE
- CAS:538-60-3
- MF:C14H15O3P
- Structure:
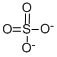
- Chemical Name:SULFATE STANDARD
- CAS:14808-79-8
- MF:O4S-2
- Chemical Name:PHOSPHODIESTERASE II
- CAS:9068-54-6
- MF:
- Structure:
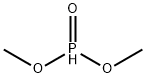
- Chemical Name:Dimethyl phosphite
- CAS:868-85-9
- MF:C2H7O3P
- Structure:
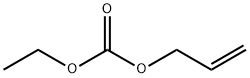
- Chemical Name:CARBONIC ACID ALLYL ETHYL ESTER
- CAS:1469-70-1
- MF:C6H10O3
- Structure:
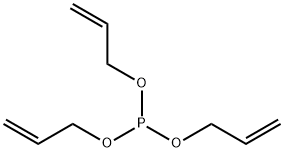
- Chemical Name:TRIALLYL PHOSPHITE
- CAS:102-84-1
- MF:C9H15O3P
- Structure:
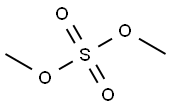
- Chemical Name:Dimethyl sulfate
- CAS:77-78-1
- MF:C2H6O4S