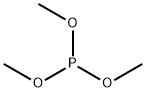
Trimethyl phosphite is a colorless liquid.
Distinctive pungent, pyridine-like odor. The Odor
Threshold is 0.0001 ppm.
Trimethyl phosphite has been used primarily as an intermediate in the manufacture of pesticides and the synthesis of organophosphate insecticides. It is also used as a fire retardants in the production of textiles, as an intermediate in the production of flame-retardant polymers for polyurethane foams, and as a catalyst.
ChEBI: Trimethyl phosphite is an organophosphorus compound that is phosphane in which the three hydrogens are replaced by methoxy groups. It is an alkylating agent used primarily in the synthesis of organophosphate compounds. It has a role as an alkylating agent.
Produced via a reaction between phosphorus trichloride and
methanol in the presence of a tertiary amine catalyst such as
diethylaniline.
The reaction of trimethyl phosphite and dimethyl acetylenedicarboxylate in the presence of dimedone as the proton source/nucleophile leads to 2-(dimethoxy-phosphoryl)-3-(2-hydroxy-4,4-dimethyl-6-oxo-cyclohex-1-enyl)-succinic acid dimethyl ester in 91 % yields.
It reacts with a catalytic amount of methyl iodide in the Arbuzov reaction to give dimethyl methylphosphonate:
P(OCH3)3 → CH3P(O)(OCH3)2
It is susceptible to oxidation to trimethyl phosphate.
Trimethyl phosphite appears as a clear colorless liquid with a strong foul odor. Flash point 99°F. Denser than water and insoluble in water. Vapors heavier than air. It is used in making pesticides, flame retardants and organophosphorous additives.
Highly flammable. Insoluble in water and denser than water. Slowly reacts with water to form phosphoric acid and corresponding organic alcohol. Trimethyl phosphite hydrolyzes in water to form dimethyl phosphite and methanol. The hydrolysis rate of trimethyl phosphite is expected to be similar to the hydrolysis rate of triethyl phosphite(SRC).
As soon as Trimethyl phosphite contacted a small amount of magnesium perchlorate in a flask, there was a flash and an explosion that shattered the flask (Allison 1968).
Trimethyl phosphite is a skin, eye, and mucous membrane irritant with low subacute inhalation toxicity. Its irritating action on rabbits’ skin was moderate to severe. The pure liquid instilled into the eyes can cause severe irritation and swelling, which can last for a few days. Chronic exposure to 300–600 ppm concentration in air produced lung inflammation and cataracts in mice. There was no acute inhalation toxicity observed in test animals. The oral and dermal toxicities were low.
LD50 value, oral (rats): 1600 mg/kg
Teratogenic effects showing gross abnormalities were observed in newborn rats when pregnant rats were dosed with high concentrations of trimethyl phosphite.
The odor threshold for this compound was determined to be 0.0001 ppm (ACGIH 1986). The odor is irritating and pungent at high concentrations..
Special Hazards of Combustion Products: Toxic fumes of PO x
Moderately toxic by
ingestion and skin contact. An experimental
teratogen. A severe skin and eye irritant.
Flammable liquid when exposed to heat,
flame, or oxidizers. To fight fire, use water,
foam, fog, CO2. Violent explosive reaction
on contact with magnesium perchlorate or
trimethyl platinum(IV) azide tetramer. When
heated to decomposition it emits toxic
fumes of POx. An intermediate in the
production of pesticides, fire retardants, and
organic phosphorus additives. See also
ESTERS.
Trimethyl phosphite is a flame
retardant, and used as an intermediate in the manufacture
of a number of pesticides, and organophosphorus
additives.
Trimethyl phosphite was genotoxic in
mouse lymphoma assays but was not mutagenic
in various bacterial assays.
UN2329 Trimethyl phosphite, Hazard Class: 3;
Labels: 3-Flammable liquid.
Treat the phosphite with Na (to remove water and any dialkyl phosphonate), then decant and distil it with protection against moisture. It has also been treated with sodium wire for 24hours, then distilled in an inert atmosphere onto activated molecular sieves [Connor et al. J Chem Soc, Dalton Trans 511 1986]. It can be fractionally distilled using a spinning band column at high reflux ratio. It is a colourless liquid which is slowly hydrolysed by H2O. [Gillis et al. J Am Chem Soc 80 2999 1958, NMR: Callis et al. J Am Chem Soc 79 2719 1957, Kosolapoff Organophosphorus Compounds, Wiley p 203 1950, Beilstein 1 IV 1256.]
Vaor May form explosive mixture with
air. Incompatible with oxidizers (chlorates, nitrates, peroxides,
permanganates, perchlorates, chlorine, bromine, fluorine,
etc.); contact may cause fires or explosions. Violent
reaction with magnesium perchlorate.
Keep away from alkaline materials, strong bases, strong
acids, oxoacids, epoxides. Reacts (hydrolyzes) with water.
Incompatible with air, moisture.
Trimethyl phosphite may be burned in a chemical incinerator equipped with an afterburner and scrubber.