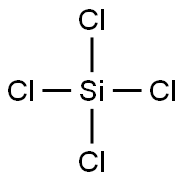
Colorless fuming liquid; suffocating odor; density 1.52 g/mL; freezes at –68.9°C; boils at 57.7°C; vapor pressure 235 torr at 25°C; critical temperature 235°C; critical pressure 35.45 atm; critical volume 326 cm3/mol; decomposes in water forming silicic acid and HCl; soluble in benzene, toluence, chloroform, and ether.
Silicon tetrachloride was first prepared by Berzelius in 1823. It is used widely in preparing pure silicon and many organosilicon compounds such as silicone. It also is used to produce smoke screens in warfare.
Silicon tetrachloride is prepared by heating silicon dioxide and carbon in a stream of chlorine:
SiO2 + C + 2Cl2 → SiCl4 + CO2
Also, the compound may be prepared by heating silicon with chlorine or dry hydrogen chloride:
Si + 2Cl2 → SiCl4
Si + 4HCl → SiCl4 + 2H2
Silicon tetrachloride decomposes in water forming silicic acid (precipitated silica) and hydrochloric acid:
SiCl4 + 3H2O → H2SiO3 + 4HCl
Reactions with alcohols yield esters of orthosilicic acid. For example, with ethanol the product is tetraethyl orthosilicate or tetraethoxysilane, Si(OC2H5)4:
SiCl4 + 4C2H5OH → Si(OC2H5)4 + 4HCl
An important class of organosilicon compounds known as silicones that are used as lubricants, resins, elastomers, and antifoaming agents in high-vacuum diffusion pumps are synthesized from silicon tetrachloride. Silicon tetrachloride reacts with Grignard reagents, RMgCl to form monoalkyltrichlorosilanes, RSiCl3, dialkyldichlorosilanes, R2SiCl2, trialkylmonochlorosilanes, R3SiCl, and tetraalkylsilanes, R4Si:
SiCl4 + RMgCl → RSiCl3 + MgCl2
SiCl4 + 2RMgCl → R2SiCl2 + 2MgCl2
SiCl4 + 3RMgCl → R3SiCl + 3MgCl2
SiCl4 + 4RMgCl → R4Si + 4MgCl2
The alkylchlorosilanes on hydrolysis form various types of silicones. For example, hydrolysis of trialkylmonochlorosilanes yields sylil ethers, R3SiOSiR3, which form silicones:
2R3SiCl + H2O → R3SiOSiR3 + 2HCl
Silicon tetrachloride reacts with diethylzinc to form tetraethylsilane. This compound was synthesized by Friedel and Crafts in 1863, the first organosilicon compound:
SiCl4 + 2Zn(C2H5)2 → Si(C2H5)4 + 2ZnCl2
Silicon tetrachloride reacts with alkyl chloride and sodium to form thesame tetraalkylsilane:
SiCl4 + 4C2H5Cl + 8Na → Si(C2H5)4 + 8NaCl
Silicon tetrachloride reacts with acetic anhydride to form silicon tetraacetate (tetraacetoxysilane). This reaction was discovered by Friedel and Ladenburg in 1867:
SiCl4 + 4(CH3CO)2O → (CH3COO)4Si + 4CH3COCl
Silicon tetraacetate can also be made by the reaction of silicon tetrachloride with sodium acetate. In general any carboxylate salt of silicon can be prepared from silicon tetrachloride by this reaction:
SiCl4 + 4CH3COO Na → (CH3COO)4Si + 4NaCl
Ladenburg in 1873 synthesized phenyltrichlorosilane, C6H5SiCl3 by heating silicon tetrachloride with diphenylmercury:
SiCl4 + (C6H5)2 Hg → C6H5SiCl3 + C6H5HgCl
Silicon tetrachloride undergoes addition with olefinic and acetylenic unsaturated hydrocarbons. In these addition reactions, one chlorine atom adds to one carbon atom of the double or triple bond while the rest of the unit —SiCl3 attaches to the other carbon atom forming a silicon—carbon bond:
SiCl4 + H2C=CH2 → ClCH2—CH2SiCl3
SiCl4 + HC≡CH → ClCH=CHSiCl3
Silicon tetrachloride is reduced to metallic silicon when heated with sodium, potassium, and a number of metals:
SiCl4 + Mg → Si + MgCl2
It reacts with carbon monoxide to form a compound with a silicon carbon bond:
SiCl4 + CO → ClC(=O)SiCl3
Reaction with excess amine forms amine derivatives of silicon:
SiCl4 + HN(CH3)2 → Si[N(CH3)2]4 + 4HN(CH3)2•HCl
The vapors are very toxic and irritating to the eyes, throat, and mucous membrane.
Chlorosilanes (general formula RnHmSiCl4-n-m, where R is an
alkyl, aryl, or olefin group) are compounds in which silicon is
bound to between one and four chlorine atoms, bonds with
hydrogen and/or organic groups making its total number of
bonds up to four. Chlorosilanes react with water, moist air, and
steam, producing heat and toxic, corrosive hydrogen chloride
fumes. Contact between gaseous hydrogen chloride and metals
may release gaseous hydrogen, which is inflammable and
explosive. Chlorosilanes react vigorously with oxidizing agents,
alcohols, strong acids, strong bases, ketones, and aldehydes.
Chlorosilanes are chemical intermediates used in the production
of silicon and silicon-containing materials, and in the
semiconductor industry; they are also protecting agents for
intermediates in pharmaceutical syntheses. The most important
industrially utilized silicon halides are trichlorosilane and
silicon tetrachloride.
Silicon tetrachloride (SiCl4) can be manufactured by chlorination
of silicon compounds such as ferrosilicon or silicon
carbide, or by heating silicon dioxide and carbon in a stream of
chlorine. It can also be obtained as a by-product in the
production of zirconium tetrachloride, and in the past
substantial quantities were produced by this route, which in
recent decades has lost importance owing to the reduced
demand for zirconium in nuclear facilities. Nowadays, industrial
silicon tetrachloride is produced either by direct reaction of
hydrogen chloride with silicon – this product mainly being
employed as an intermediate in fumed silica production – or as
the by-product of the production of silane for the microelectronics
industry by disproportionation of trichlorosilane.
Silicon tetrachloride (SiCl4), produced when both silicon and chlorine are combined at high
temperatures, is used by the military to produce smoke screens.
. Silicon tetrachloride (SiCl4) may be used as an intermediate in the manufacture of high purity silicon. High purity silicon derived from silicon tetrachloride may find major applications in the semiconductors industry and photovoltaic cells.
. High purity SiCl4 may be used to manufacture of optical fibers.
Tetrachlorosilane, can be used as a coupling agent for the synthesis of amine from carboxylic acid and an amide. It can also be used in preparation of high purity silicon, used in photovoltaic cells, and in the semiconductors industry.
Manufactured directly by the reaction of chlorine on silicon
metal or ferrosilicon at 500�C or silicon carbide.
Tetrachlorosilane is a colorless, fuming liquid with a pungent odor. Tetrachlorosilane is decomposed by water to hydrochloric acid with evolution of heat. Tetrachlorosilane is corrosive to metals and tissue in the presence of moisture. Tetrachlorosilane is used in smoke screens, to make various silicon containing chemicals, and in chemical analysis.
Chlorosilanes, such as Tetrachlorosilane, are compounds in which silicon is bonded to from one to four chlorine atoms with other bonds to hydrogen and/or alkyl groups. Chlorosilanes react with water, moist air, or steam to produce heat and toxic, corrosive fumes of hydrogen chloride. They may also produce flammable gaseous H2. They can serve as chlorination agents. Chlorosilanes react vigorously with both organic and inorganic acids and with bases to generate toxic or flammable gases. Tetrachlorosilane is incompatible with alkali metals and dimethyl sulfoxide.
Toxic by ingestion and inhalation, strong
irritant to tissue.
Inhalation causes severe irritation of upper respiratory tract resulting in coughing, choking, and a feeling of suffocation; continued inhalation may produce ulceration of the nose, throat, and larynx; if inhaled deeply, edema of the lungs may occur. Contact of liquid with eyes causes severe irritation and painful burns; may cause permanent visual impairment. Liquid may cause severe burns of skin. Repeated skin contact with dilute solutions or exposure to concentrated vapors may cause dermatitis. Ingestion causes severe internal injury with pain in the throat and stomach, intense thirst, difficulty in swallowing, nausea, vomiting, and diarrhea; in severe cases, collapse and unconsciousness may result.
Behavior in Fire: Contact with water in foam applied to adjacent fires will produce irritating fumes of hydrogen chloride.
Flammability and Explosibility
Not classified
Mildly toxic by inhalation. A corrosive irritant to eyes, skin, and mucous membranes. Reacts with water to form HCl. Violent reaction with Na, K. When heated to decomposition it emits toxic fumes of Cl-. See also CHLOROSILANES.
Studies of rats subjected to acute inhalation of 10 structurally
similar chlorosilanes, including tetrachlorosilane, suggest that
the acute toxicity of chlorosilanes is largely due to the hydrogen
chloride hydrolysis product. The observed effects were similar
to those of HCl inhalation both qualitatively (clinical signs)
and quantitatively (molar equivalents of hydrogen chloride at
the atmospheric LC50).
Distil it under vacuum and store it in sealed ampoules under N2. It fumes in moist air and is very sensitive to moisture. It is soluble in organic solvents. It is a strong irritant. [Schenk in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I pp 682-683 1963.]
Silicon tetrachloride is a colorless, noninflammable, volatile
liquid with a pungent, suffocating odor. It fumes in air and is
corrosive to metals and tissues in the presence of moisture. In
experiments at Argonne National Laboratory in which it was
mixed with water and stirred under room conditions, about
35% of the theoretical yield of HCl evolved as a gas in the first
minute. It also reacts very rapidly with alcohols, primary and
secondary amines, ammonia, and other compounds containing
active hydrogen atoms. Thermal decomposition or burning
may produce dense white clouds of silicon oxide particles and
hydrogen chloride.
Silicon tetrachloride is a by-product in the production of
polysilicon, the key component of sunlight-capturing wafers in
solar energy panels, and for each ton of polysilicon produced,
at least four tons of silicon tetrachloride liquid waste are
generated. Pollution by silicon tetrachloride has been reported
in China, associated with the increased demand for photovoltaic
cells that has been stimulated by subsidy programs.
The initial threshold screening level (ITSL) for silicon tetrachloride (CAS #10026-04-7) is 20 μg/m3 based on an annual averaging time. A second acute ITSL for silicon tetrachloride of 2,000 μg/m3 based on a 1-hour averaging time.