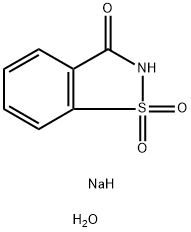
Saccharin sodium dihydrate is the sodium dihydrate form of saccharin, which belongs to the family of aromatic homomonocyclic compounds. It is an artificial sweetener that is hundreds times as sweet as sucrose. It is mainly used as a food additive to sweeten many products such as drinks, candies, cookies, and medicine. Saccharin is relatively safe for human beings without providing food energy and any nutritional value. It is also safe for diabetes patients to consume.
Saccharin sodium occurs as a white, odorless or faintly aromatic,
efflorescent, crystalline powder. It has an intensely sweet taste, with
a metallic or bitter aftertaste that at normal levels of use can be
detected by approximately 25% of the population. The aftertaste
can be masked by blending saccharin sodium with other sweeteners.
Saccharin sodium can contain variable amounts of water.
Saccharin Sodium Salt Dihydrate is a non-nutritive sweetener; pharmaceutic aid (flavor). Saccharin was formerly listed as reasonably anticipated to be a human carcinogen; delisted because the cancer data are not sufficient to meet the current criteria for this listing.
Sweetener (non-nutritive).
Saccharin is produced by the oxidation of o-toluene sulfonamide by
potassium permanganate in a solution of sodium hydroxide.
Acidification of the solution precipitates saccharin, which is then
dissolved in water at 50℃ and neutralized by addition of sodium
hydroxide. Rapid cooling of the solution initiates crystallization of
saccharin sodium from the liquors.
Pharmaceutical Applications
Saccharin sodium is an intense sweetening agent used in beverages,
food products, table-top sweeteners, and pharmaceutical formulations
such as tablets, powders, medicated confectionery, gels,
suspensions, liquids, and mouthwashes. It is also used
in vitamin preparations.
Saccharin sodium is considerably more soluble in water than
saccharin, and is more frequently used in pharmaceutical formulations.
Its sweetening power is approximately 300–600 times that of
sucrose. Saccharin sodium enhances flavor systems and may be used
to mask some unpleasant taste characteristics.
Injection of saccharin sodium has been used to measure the armto-
tongue circulation time.
There has been considerable controversy concerning the safety of
saccharin and saccharin sodium in recent years; however, it is now generally regarded as a safe, intense sweetener. See Saccharin for
further information.
The WHO has set a temporary acceptable daily intake of up to
2.5 mg/kg body-weight for saccharin, including its salts.(3) In the
UK, the Committee on Toxicity of Chemicals in Food, Consumer
Products, and the Environment (COT) has set an acceptable daily
intake for saccharin and its salts (expressed as saccharin sodium) at
up to 5 mg/kg body-weight.
LD50 (mouse, oral): 17.5 g/kg
LD50 (rat, IP): 7.1 g/kg
LD50 (rat, oral): 14.2 g/kg
Saccharin sodium is stable under the normal range of conditions
employed in formulations. Only when it is exposed to a high
temperature (125℃) at a low pH (pH 2) for over 1 hour does
significant decomposition occur. The 84% grade is the most stable
form of saccharin sodium since the 76% form will dry further under
ambient conditions. Solutions for injection can be sterilized by
autoclave.
Saccharin sodium should be stored in a well-closed container in a
dry place.
Saccharin sodium does not undergo Maillard browning.
Accepted for use as a food additive in Europe; ‘E954’ is applied to
both saccharin and saccharin salts. Included in the FDA Inactive
Ingredients Database (buccal and dental preparations; IM and IV
injections; oral and topical preparations). Included in nonparenteral
medicines licensed in the UK. Included in the Canadian List of
Acceptable Non-medicinal Ingredients.
https://en.wikipedia.org/wiki/Saccharin
https://pubchem.ncbi.nlm.nih.gov/compound/saccharin#section=Top