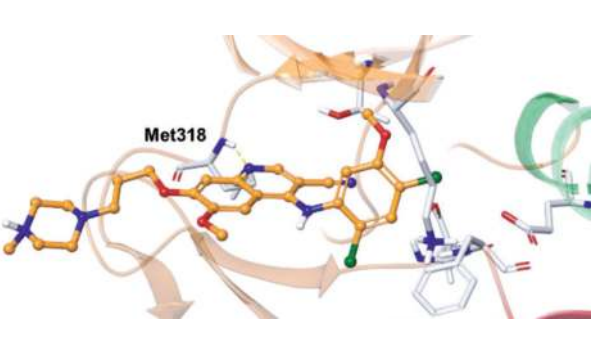
In the co-crystal structures of the two molecules of dasatinib (green and blue) in the asymmetric unit cell with ABL, it binds to an active DFG-information of ABL. The aminothiazole core, which is orthogonal to the 2-chloro-6-methylphenyl moiety, occupies the site that normally binds the adenine group of ATP. The pyrimidine NH and the thiazole nitrogen interact with the backbone amide carbonyl and NH of the hinge region Met318, and the carboxamide NH forms an H-bond with the side chain hydroxyl of Thr315. The terminal hydroxyethyl group interacts differently with each of the two molecules in the asymmetric unit cell: (1) via a hydrogen bond between the hydroxyethyl group and the backbone carbonyl of Tyr320; and (2) with the hydroxyethyl group pointing in the opposite direction and approaching the solvent front. By binding strongly to the active conformation of ABL within the ATP-binding site (vs. the inactive conformation for imatinib), dasatinib efficiently competes with ATP. Due to a different binding mode from that of imatinib, it maintains cellular activity against most imatinib-resistant BCR-ABL mutations (but not the T315I mutation due to the loss of a hydrogen bond with T315 and increased steric clash with the bulky isobutyl chain of 315I). Unfortunately, binding to the more highly conserved active conformation results in reduced selectivity relative to imatinib, and as a result, dasatinib inhibits many additional protein kinases at physiologically relevant concentrations.
Bosutinib, similar to dasatinib, was developed as a dual inhibitor of the SCR family of kinases and the ABL kinase, thus applicable for Ph+ leukemias. In September 2012, the US FDA approved bosutinib (also referred to as SKI-607) for the treatment of relapsed or refractory chronicmyeloid leukemia (CML) for patients with resistance or intolerance to prior therapy.
Pale Yellow Solid
Class: non-receptor tyrosine kinase
Treatment: CML
Oral bioavailability = 34%
Elimination half-life = 22.5 h
Protein binding = 95%
A novel Src kinase inhibitor, suppresses migration and invasion of human breast cancer cells.
Bosutinib (SKI-606) is a novel, dual Src/Abl inhibitor with IC50 of 1.2 nM and 1 nM, respectively. It is built around a chemical scaffold that is different from that of imatinib. peripheral vasolidator increases cerebral blood flow, potential therapeutic for Altzheimer’s disease, cognitive impairment and dementia.
ChEBI: Bosutinib is an aminoquinoline that is 4-[(2,4-dichloro-5-methoxyphenyl)amino]-7-[3-(4-methylpiperazin-1-yl)propoxy]quinoline bearing additional cyano and methoxy substituents at positions 3 and 6 respectively. It has a role as an antineoplastic agent and a tyrosine kinase inhibitor. It is a nitrile, a N-methylpiperazine, an aromatic ether, a tertiary amino compound, an aminoquinoline and a dichlorobenzene.
Bosutinib is a safe oral medicine with good bioavailability. It is effective for treating chronic myeloid leukemia (CML). It is also a potent ATP-competitive inhibitor for Src tyrosine kinase. Bosutinib inhibits epidermal growth factor receptor (EGFR). It suppresses solid tumors associated with breast, pancreas and prostate.
Bosutinib (SKI-606) is an orally active; dual Src/Abl tyrosine kinase inhibitor with potent antiproliferative activity. It does not appear to inhibit c-Kit and PDGRF, which are thought to be the cause of numerous side effects in anticancer treatment with some other tyrosine kinase inhibitors.
Protein kinase inhibitor:
Treatment of Philadelphia chromosome-positive
chronic myelogenous leukaemia resistant or
intolerant to prior therapy
Possible side effects of bosutinib include Nausea, vomiting, stomach/abdominal pain, loss of appetite, cough, joint pain, headache, or dizziness may occur. If any of these effects last or get worse, tell your doctor or pharmacist promptly. Diarrhea is a common side effect.
www.webmd.com
Potentially hazardous interactions with other drugs
Analgesics: possibly increased risk of ventricular
arrhythmias with methadone.
Anti-arrhythmics: possibly increased risk of ventricular
arrhythmias with amiodarone and disopyramide;
concentration possibly increased by dronedarone -
avoid or consider reducing dose of bosutinib.
Antibacterials: concentration possibly increased
by ciprofloxacin, clarithromycin, erythromycin and
telithromycin - avoid or consider reducing dose
of bosutinib; possibly increased risk of ventricular
arrhythmias with moxifloxacin; concentration
reduced by rifampicin and possibly rifabutin - avoid.
Antidepressants: concentration possibly reduced by
St John’s wort - avoid.
Antiepileptics: concentration possibly reduced
by carbamazepine, fosphenytoin, phenobarbital,
phenytoin and primidone - avoid.
Antifungals: concentration increased by ketoconazole
and possibly by fluconazole, itraconazole,
posaconazole and voriconazole - avoid or consider
reducing dose of bosutinib.
Antimalarials: possibly increased risk of
ventricular arrhythmias with chloroquine and
hydroxychloroquine.
Antipsychotics: possibly increased risk of ventricular
arrhythmias with haloperidol; avoid with clozapine,
increased risk of agranulocytosis.
Antivirals: concentration possibly increased by
atazanavir, boceprevir, darunavir, fosamprenavir,
indinavir, ritonavir, saquinavir and telaprevir - avoid
or consider reducing dose of bosutinib; concentration
possibly reduced by efavirenz and etravirine - avoid.
Aprepitant: concentration possibly increased - avoid
or consider reducing dose of bosutinib.
Beta-blockers: possibly increased risk of ventricular
arrhythmias with sotalol.
Bosentan: concentration of bosutinib possibly
reduced - avoid.
Calcium channel blockers: concentration possibly
increased by diltiazem or verapamil - avoid or
consider reducing dose of bosutinib.
Cytotoxics: concentration possibly increased by
imatinib - avoid or consider reducing dose of bosutinib.
Domperidone: avoid concomitant use, risk of
ventricular arrhythmias.
Fosaprepitant: concentration possibly increased by
fosaprepitant - avoid or consider reducing dose of
bosutinib
Grapefruit juice: concentration possibly increased by
grapefruit juice - avoid or consider reducing dose of
bosutinib.
Modafinil: concentration of bosutinib possibly
reduced - avoid.
Mainly hepatically metabolised. The major circulating
metabolites identified in plasma are oxydechlorinated
(M2) bosutinib (19% of parent exposure) and
N-desmethylated (M5) bosutinib (25% of parent
exposure), with bosutinib N-oxide (M6) as a minor
circulating metabolite. All the metabolites are inactive.
Excretion is mainly via the faeces.
Bosutinib is a synthetic quinolone derivative and dual kinase inhibitor that targets both Abl and Src kinases with potential antineoplastic activity. Unlike imatinib, bosutinib inhibits the autophosphorylation of both Abl and Src kinases, resulting in inhibition of cell growth and apoptosis. Because of the dual mechanism of action, this agent may have activity in resistant CML disease, other myeloid malignancies and solid tumors. Abl kinase is upregulated in the presence of the abnormal Bcr-abl fusion protein which is commonly associated with chronic myeloid leukemia (CML). Overexpression of specific Src kinases is also associated with the imatinib-resistant CML phenotype.
[1] JENNIFER M GOLAS. SKI-606, a 4-anilino-3-quinolinecarbonitrile dual inhibitor of Src and Abl kinases, is a potent antiproliferative agent against chronic myelogenous leukemia cells in culture and causes regression of K562 xenografts in nude mice.[J]. Cancer research, 2003, 63 2: 375-381.
[2] L L REMSING RIX. Global target profile of the kinase inhibitor bosutinib in primary chronic myeloid leukemia cells[J]. Leukemia, 2008, 23 3: 477-485. DOI:
10.1038/leu.2008.334[3] SARA REDAELLI. Activity of bosutinib, dasatinib, and nilotinib against 18 imatinib-resistant BCR/ABL mutants.[J]. Journal of Clinical Oncology, 2009, 27 3: 469-471. DOI:
10.1200/jco.2008.19.8853[4] ROBERT J MACDONALD. Src family kinase inhibitor bosutinib enhances retinoic acid-induced differentiation of HL-60 leukemia cells.[J]. Leukemia & Lymphoma, 2018, 59 12: 2941-2951. DOI:
10.1080/10428194.2018.1452213[5] ADINA VULTUR. SKI-606 (bosutinib), a novel Src kinase inhibitor, suppresses migration and invasion of human breast cancer cells.[J]. Molecular Cancer Therapeutics, 2008, 7 5: 1185-1194. DOI:
10.1158/1535-7163.mct-08-0126