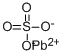White monoclinic or rhombic crystals; refractive index 1.877; density 6.20 g/cm3; melts at 1,170°C; very slightly soluble in water, 42.5 mg/L at 25°C; KSP1.82x10–8; sparingly soluble in hydrochloric and nitric acids; slightly soluble in dilute sulfuric acid, more soluble in concentrated sulfuric acid; insoluble in alcohol; soluble in caustic soda and ammonium salts solutions.
Lead sulfate occurs in nature as the mineral, anglesite. It is an essential component of lead-sulfuric acid storage batteries and forms during discharge cycles. It is used as an additive to stabilize clay soil for construction. Other applications are in photography and as a pigment.
Lead sulfate precipitates when an aqueous solution of lead nitrate or other soluble salt of lead is treated with sulfuric acid.
Alternatively, lead sulfate may be prepared by treating lead oxide, hydroxide, or carbonate with concentrated sulfuric acid under warm condition. It also is produced when lead sulfide is roasted in air.
Low to moderate toxicity by ingestion. Irritant action on eye, skin, and mucous membranes.
Lead sulfate is a heavy, white crystallinepowder. Molecular weight=303.25; Freezing/Meltingpoint = 1170℃. Hazard Identification (based on NFPA-704M Rating System): Health 3, Flammability 0, Reactivity 1.Soluble in water.
Lead sulfate is a heavy, white crystalline powder.
It has been used in photography in combination with silver bromide and is used in the stabilization of clay soil for adobe structures, earth-fill dams, and roads.
Lead(II) sulfate is used in pigments, galvanic batteries, lithography, weighting fabrics. It is also used as intermediates.
As pigment instead of white lead; manufacture of galvanic and lead-acid batteries; manufacture of minium, in lithography; preparing rapidly drying oil varnishes; weighting fabrics.
Lead sulfate, PbSO4, is soluble in concentrated acids and alkalies, forming hydroxyplumbites. It is prepared by treating lead oxide, hydroxide, or carbonate with warm sulfuric acid, or by treating a soluble lead salt with sulfuric acid.
anglesite: A mineral form oflead(II) sulphate, PbSO4.
A white crystalline solid. Insoluble in water and sinks in water. Contact may irritate skin, eyes, and mucous membranes. May be mildly toxic by ingestion, inhalation and skin absorption. Used to make other chemicals. Use: in lithography, battery acid solution treated fabrics, used in varnishes.
LEAD(II) SULFATE reacts explosively with boric acid [Bretherick 1979. p. 1029]. Special Hazards of Combustion Products: Toxic metal fumes [USCG, 1999].
INHALATION: Joint and muscle pains, headache, dizziness and insomnia. Weakness, frequently of extensor muscles of hand and wrist (unilateral or bilateral). Heavy contamination. Brain damage. Stupor progressing to coma - with or without convulsion, often death. Excitation, confusion, and mania less common. Cerebrospinal pressure may be increased. EYES: Caused a moderat purulent reaction and general inflammation of the rabbit eye. INGESTION: Abdominal pain, diarrhea, constipation, loss of appetite, muscular weakness, headache, blue line on gums, metallic taste, nausea and vomiting.
Special Hazards of Combustion Products: Toxic metal fumes
reagent type: catalyst
core: lead
Lead sulfate is used in storage batteries and paint pigments. Used in the making of alloys, fastdrying oil varnishes; weighting fabrics; in lithography
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Do not induce vomiting.Note to physician: whole blood lead levels, circulatingplasma/erythrocyte lead concentration ratio, urine ALA,and erythrocyte protoporphyrin fluorescent microscopy mayall be useful in monitoring or assessing lead exposure.Chelating agents, such as edetate disodium calcium (CaEDTA) and penicillamine (not penicillin), are generallyuseful in the therapy of acute lead intoxication.Antidotes and special procedures for lead: Persons with significant lead poisoning are sometimes treated with CaEDTA while hospitalized. This “chelating” drug causes arush of lead from the body organs into the blood and kidneys, and thus has its own hazards, and must be administered only by highly experienced medical personnel undercontrolled conditions and careful observation. Ca EDTA orsimilar drugs should never be used to prevent poisoningwhile exposure continues or without strict exposure control,as severe kidney damage can result.Note to physician: For severe poisoning BAL [British AntiLewisite, dimercaprol, dithiopropanol (C3H8OS2)] has beenused to treat toxic symptoms of certain heavy metals poisoning. In the case of lead poisoning it may have SOMEvalue. Although BAL is reported to have a large margin of safety, caution must be exercised, because toxic effects maybe caused by excessive dosage. Most can be prevented bypremedication with 1-ephedrine sulfate (CAS: 134-72-5).
Color Code—White: Corrosive or Contact Hazard;Store separately in a corrosion-resistant location. Prior toworking with this chemical you should be trained on its proper handling and storage. Lead sulfate must be stored toavoid contact with oxidizers (such as perchlorates, peroxides, permanganates, chlorates, and nitrates) and chemicallyactive metals (such as potassium, sodium, magnesium, andzinc), since violent reactions occur. Lead is regulated by anOSHA Standard 1910.1025. All requirements of the standard must be followed. A regulated, marked area should beestablished where this chemical is handled, used, or storedin compliance with OSHA Standard 1910.1045.
UN2291 Lead compounds, soluble n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical
Name Required. UN1794 Lead sulfate with more than 3%
free acid, Hazard class: 8; Labels: 8-Corrosive material.
UN3077 Environmentally hazardous substances, solid, n.o.
s., Hazard class: 9; Labels: 9-Miscellaneous hazardous
material, Technical Name Required
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions. Contact with potassium can cause explosion. Keep
away from alkaline materials, strong bases, strong acids,
oxoacids, and epoxides