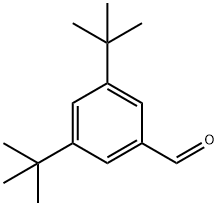
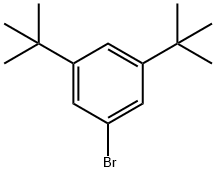
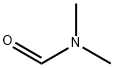
1-Bromo-3,5-di-tert-butylbenzene (5.00 g, 18.57 mmol) was dissolved in tetrahydrofuran (THF, 50 mL) under nitrogen protection. The reaction mixture was cooled to -78°C. n-Butyllithium (2.5 M hexane solution, 22.29 mL, 55.72 mmol) was slowly added, keeping the temperature at -78°C. The reaction mixture was stirred for 30 min at -78°C. The reaction mixture was then stirred for 30 min at -78°C. Subsequently, N,N-dimethylformamide (DMF, 4.31 mL, 55.72 mmol) was added dropwise. The reaction mixture was slowly warmed to 0°C and stirred at this temperature for 2.5 hours. Upon completion of the reaction, the reaction was quenched by the addition of aqueous ammonium chloride solution (30 mL). The reaction mixture was extracted with ethyl acetate (EtOAc, 3 x 20 mL). The organic phases were combined, dried over anhydrous sodium sulfate (Na2SO4), filtered, the filtrate collected and concentrated under reduced pressure. The residue was purified by fast column chromatography using a 0-10% ethyl acetate/petroleum ether gradient elution to afford 3,5-bis(tert-butyl)benzaldehyde (2.96 g, 73% yield) as a white solid.LC-ES/MS m/z 219 [M + H]+.