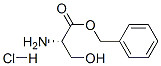
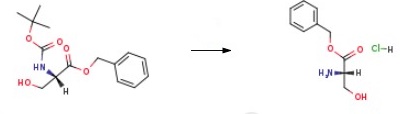
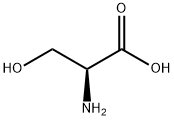
a) To a 250 mL three-necked flask was added a mixed solution of benzyl alcohol (70 mL) and carbon tetrachloride (70 mL) of L-serine (15.00 g, 0.14 mol) and p-toluenesulfonic acid (29.30 g, 0.15 mol). The reaction mixture was heated to reflux overnight under a Dean-Stark apparatus (a total of 7 mL of water was collected). Upon completion of the reaction, carbon tetrachloride was removed by distillation under reduced pressure. Dichloromethane (50 mL) was added to the residue and the resulting solution was washed sequentially with saturated aqueous sodium bicarbonate (3 x 50 mL) and 1 M hydrochloric acid (3 x 50 mL). The aqueous phases were combined, concentrated under reduced pressure and dried to give L-serine benzyl ester hydrochloride (17.52 g, 54% yield) as colorless crystals with melting point 169-171 °C. LC-MS m/z: 196.15 [M+H]+.
(b) Boc-phenylalanine (2.92 g, 11.00 mmol) was dissolved in N,N-dimethylformamide (50 mL) in a dry 100 mL three-necked flask. N,N-diisopropylethylamine (1.88 mL, 11.00 mmol), 1-ethyl-(3-dimethylaminopropyl)carbodiimide hydrochloride (1.95 mL, 11.00 mmol), and 1-hydroxybenzotriazole (1.49 g, 11.00 mmol) were added sequentially. After stirring at room temperature for 5 minutes, L-serine benzyl ester hydrochloride (2.31 g, 10.00 mmol) was added and the reaction was continued with stirring overnight. After completion of the reaction, the reaction mixture was diluted with ether (150 mL) and washed sequentially with 1 M hydrochloric acid (2 x 100 mL), saturated saline (100 mL), saturated aqueous sodium bicarbonate solution (2 x 100 mL) and saturated saline (100 mL). The organic phase was dried over anhydrous magnesium sulfate and concentrated under reduced pressure. The target product (2.08 g, 78% yield) was purified by fast column chromatography using ethyl acetate/hexane (1:1, v/v) as eluent to afford the target product (2.08 g, 78% yield) as a colorless oil.