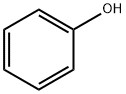
Фенол
- английское имяPhenol
- CAS №108-95-2
- CBNumberCB4362168
- ФормулаC6H6O
- мольный вес94.11
- EINECS203-632-7
- номер MDLMFCD00002143
- файл Mol108-95-2.mol
| Температура плавления | 40-42 °C(lit.) |
| Температура кипения | 182 °C(lit.) |
| плотность | 1.071 g/mL at 25 °C(lit.) |
| Плотность накопления | 620kg/m3 |
| плотность пара | 3.24 (vs air) |
| давление пара | 0.09 psi ( 55 °C) |
| FEMA | 3223 | PHENOL |
| показатель преломления | n |
| Fp | 175 °F |
| температура хранения | 2-8°C |
| растворимость | H2O: 50 mg/mL at 20 °C, clear, colorless |
| форма | liquid |
| пка | 9.89(at 20℃) |
| Удельный вес | 1.071 |
| цвет | faintly yellow |
| РН | 6.47(1 mM solution);5.99(10 mM solution);5.49(100 mM solution); |
| Запах | Sweet, medicinal odor detectable at 0.06 ppm |
| Порог?обнаружения?запаха? | 0.0056ppm |
| Пределы взрываемости | 1.3-9.5%(V) |
| Биологические источники | synthetic |
| Odor Type | phenolic |
| Растворимость в воде | 8 g/100 mL |
| Точка замерзания | 41℃ |
| Чувствительный | Air & Light Sensitive |
| Мерк | 14,7241 |
| Номер JECFA | 690 |
| БРН | 969616 |
| констант закона Генри | 1.09 at 5 °C (average derived from six field experiments, Lüttke and Levsen, 1997) |
| Пределы воздействия | TLV-TWA skin 5 ppm (~19 mg/m3 ) (ACGIH, MSHA, and OSHA); 10-hour TWA 5.2 ppm (~20 mg/m3 ) (NIOSH); ceiling 60 mg (15 minutes) (NIOSH); IDLH 250 ppm (NIOSH). |
| Диэлектрическая постоянная | 4.3(10℃) |
| Стабильность | Hygroscopic |
| ИнЧИКей | ISWSIDIOOBJBQZ-UHFFFAOYSA-N |
| LogP | 1.47 at 30℃ |
| Справочник по базе данных CAS | 108-95-2(CAS DataBase Reference) |
| Вещества, добавляемые в пищу (ранее EAFUS) | PHENOL |
| FDA 21 CFR | 175.105; 175.300; 310.545 |
| Рейтинг продуктов питания EWG | 4-7 |
| FDA UNII | 339NCG44TV |
| Словарь онкологических терминов NCI | carbolic acid; phenol |
| Код УВД | C05BB05,D08AE03,N01BX03,R02AA19 |
| МАИР | 3 (Vol. 47, 71) 1999 |
| Справочник по химии NIST | Phenol(108-95-2) |
| Система регистрации веществ EPA | Phenol (108-95-2) |
| Поглощение | cut-off at 294nm in H2O at 0.5M |
| UNSPSC Code | 41116107 |
| NACRES | NA.52 |
| Коды опасности | T,C,F,Xn | |||||||||
| Заявления о рисках | 23/24/25-34-48/20/21/22-68-40-39/23/24/25-11-36-20/21/22-24/25 | |||||||||
| Заявления о безопасности | 26-36/37/39-45-36/37-28A-28-24/25-1/2-36-16-7 | |||||||||
| РИДАДР | UN 2821 6.1/PG 2 | |||||||||
| OEB | A | |||||||||
| OEL | TWA: 5 ppm (19 mg/m3), Ceiling: 15.6 ppm (60 mg/m3) [15-minute] [skin] | |||||||||
| WGK Германия | 2 | |||||||||
| RTECS | SJ3325000 | |||||||||
| F | 8-23 | |||||||||
| Температура самовоспламенения | 715 °C | |||||||||
| TSCA | Yes | |||||||||
| Класс опасности | 6.1 | |||||||||
| Группа упаковки | II | |||||||||
| кода HS | 29071100 | |||||||||
| Банк данных об опасных веществах | 108-95-2(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 orally in rats: 530 mg/kg (Deichmann, Witherup) | |||||||||
| ИДЛА | 250 ppm | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)




-
сигнальный язык
опасность
-
вредная бумага
H301+H311+H331:Токсично при проглатывании, при контакте с кожей или при вдыхании.
H341:Предполагается, что данное вещество вызывает генетические дефекты.
H411:Токсично для водных организмов с долгосрочными последствиями.
H314:При попадании на кожу и в глаза вызывает химические ожоги.
H373:Может поражать органы (Нервная система) в результате многократного или продолжительного воздействия при вдыхании.
-
оператор предупредительных мер
P260:Не вдыхать газ/ пары/ пыль/ аэрозоли/ дым/ туман.
P273:Избегать попадания в окружающую среду.
P280:Использовать перчатки/ средства защиты глаз/ лица.
P303+P361+P353:ПРИ ПОПАДАНИИ НА КОЖУ (или волосы): Снять/удалить немедленно всю загрязненную одежду. Промыть кожу водой.
P304+P340+P310:ПРИ ВДЫХАНИИ: Свежий воздух, покой. Немедленно обратиться за медицинской помощью.
P305+P351+P338:ПРИ ПОПАДАНИИ В ГЛАЗА: Осторожно промыть глаза водой в течение нескольких минут. Снять контактные линзы, если Вы ими пользуетесь и если это легко сделать. Продолжить промывание глаз.
Фенол химические свойства, назначение, производство
Описание
Phenol is a stable chemical substance and appear as colourless/white crystals with a characteristic, distinct aromatic/acrid odour. It is reactive and incompatible with strong oxidising agents, strong bases, strong acids, alkalis, and calcium hypochlorite. Phenol is flammable and may discolour in light. Phenol is used in the manufacture or production of explosives, fertiliser, coke, illuminating gas, lampblack, paints, paint removers, rubber, perfumes, asbestos goods, wood preservatives, synthetic resins, textiles, drugs, and pharmaceutical preparations. It is also extensively used as a disinfectant in the petroleum, leather, paper, soap, toy, tanning, dye, and agricultural industries.Химические свойства
Phenol, C6H5OH, also known as carbolic acid and phenylic acid, is a white poisonous crystalline solid that melts at 43 °C (110 OF) and boils at 182°C (360 OF). Phenol has a sharp burning taste,a distinctive odor, and it irritates tissue. It is toxic not only by ingestion or inhalation, but also by skin absorption. Phenol is soluble in water,alcohol,and ether. It is used in the production of resins,germicides,weedkillers,pharmaceuticals, and as a solvent in the refining of lubricating oils.Физические свойства
Phenol is a colorless or white crystalline solid that is slightly soluble in water. Phenol is the simplest of the large group of organic chemicals known as phenols, which consist of compounds where a carbon in the phenyl aromatic group (C6H5) is directly bonded to hydroxyl, OH.Вхождение
It is reported found in over 150 natural products including apricot, sour cherry, black currant, bilberry, cranberry, other berries, grapes, guava fruit, peach, pineapple, asparagus, onion, cooked potato, tomato, cinnamon bark, cassia leaf, ginger, pennyroyal oil, many cheeses, butter, milk, milk powder, boiled egg, fish and fish oil, cooked and cured meats, beer, wheaten bread, crisp bread, cognac, rose wine, cocoa, coffee, tea, whiskies, roasted filbert, roasted peanut, soybean, pecans, honey, avocado, Arctic bramble, passion fruit, beans, mushrooms, burley tobacco, cooked beef and chicken, fermented soy sauce, trassi, roasted almonds, sesame seed, fenugreek, mango, tamarind, Brazil nut, rice, rhubarb, licorice, buckwheat, watercress, malt, wort, dried bonito, loquat, myrtle berry, rosemary, Tahiti and Bourbon vanilla, endive, shrimp, crab, crayfish, clam, squid, truffle and Chinese quince.История
Phenol’s first prominent use was by Joseph Lister (1827–1912) as an antiseptic. Throughout human history,infection often resulted in death,even when the wound could be surgically treated.A broken bone piercing the skin, which today is a painful but not life-threatening injury,historically resulted in infection and possible amputation or death. Lister was inspired by Louis Pasteur’s (1822–1895) germ theory of disease,and he began to use antiseptic methods during routine surgery during the 1860s.Использование
phenol is frequently used for medical chemical face peels. It may trap free radicals and can act as a preservative. Phenol, however, is an extremely caustic chemical with a toxicity potential. It is considered undesirable for use in cosmetics. even at low concentrations, it frequently causes skin irritation, swelling, and rashes.Определение
ChEBI: An organic hydroxy compound that consists of benzene bearing a single hydroxy substituent. The parent of the class of phenols.Подготовка
Phenol is formed in dry distillation of wood, peat and coal; coal tar is one of the commercial sources of phenol and its homologues.Показания
Phenol in dilute solution (0.5% to 2%) decreases itch by anesthetizing the cutaneous nerve endings. Phenol should never be used on pregnant women or infants younger than 6 months of age.Методы производства
Historically, phenol was produced by the distillation of coal tar. Today, phenol is prepared by one of several synthetic methods, such as the fusion of sodium benzenesulfonate with sodium hydroxide followed by acidification; the hydrolysis of chlorobenzene by dilute sodium hydroxide at high temperature and pressure to give sodium phenate, which on acidification liberates phenol (Dow process); or the catalytic vapor-phase reaction of steam and chlorobenzene at 500°C (Raschig process).Всемирная организация здравоохранения(ВОЗ)
Phenol became widely used as an antiseptic following demonstration of its germicidal activity in 1867. It is an intensely corrosive substance and percutaneous absorption can produce serious systemic toxicity. It has been withdrawn from pharmaceutical preparations by at least one national regulatory authority. However, it is still used widely in concentrations of the order of 1.4% in proprietary preparations for the relief of soreness of the mouth and throat.Общее описание
A solid melting at 110°F. Colorless if pure, otherwise pink or red. Flash point 175°F. Density 9.9 lb / gal. Vapors are heavier than air Corrosive to the skin (turning skin white) but because of its anesthetic quality numbs rather than burn. Lethal amounts can be absorbed through the skin. Used to make plastics and adhesives.Реакции воздуха и воды
Decomposes slowly in air. Mixtures of 9-10% phenol in air are explosive. Soluble in waterПрофиль реактивности
PHENOL is a weak acid. Reacts exothermically with bases. Reacts with strong oxidizing agents. Emits acrid smoke and irritating fumes when heated to decomposition. Undergoes, in the presence of aluminum chloride, potentially explosive reactions with nitromethane, butadiene, formaldehyde, peroxodisulfuric acid, peroxosulfuric acid, and sodium nitrite . Reacts violently with sodium nitrate in the presence of trifluoroacetic acid [Bretherick, 5th ed., 1995, p. 770]. May corrode lead, aluminum and its alloys, certain plastics, and rubber. Phenol may explode in contact with peroxodisulfuric acid (Dns, J. Ber., 1910, 43, 1880; Z. Anorg. Chem., 1911, 73, 1911.) or peroxomonosulfuric acid. (Sidgwick, 1950, 939)Угроза здоровью
Phenol and its vapors are corrosive to the eyes, skin, and respiratory tract. The corrosive effect on skin and mucous membranes is due to a protein-degenerating effect. Repeated or prolonged skin contact with phenol may cause dermatitis, and potentially second and third-degree burns. Inhalation of phenol vapor may cause lung edema. Phenol may adversely effect the central nervous system and heart. Long-term, or repeated exposure, to phenol may have harmful effects on the liver and kidneys.While there is no evidence that phenol causes cancer in humans it is readily absorbed through the skin; systemic poisoning can occur in addition to the local caustic burns. Resorptive poisoning by a large quantity of phenol can occur even with only a small area of skin, rapidly leading to paralysis of the central nervous system and a severe drop in body temperature. Phenol is also a reproductive toxin causing increased risk of abortion and low birth weight indicating retarded development in utero.
Chemical burns from skin exposures can be decontaminated by washing with polyethylene glycol or isopropyl alcohol; flushing with copious amounts of water will help to remediate the burn. Removal of contaminated clothing is required, as well as immediate hospital treatment for large splashes.
https://ehs.ucsc.edu/lab-safety-manual/specialty-chemicals/phenol.html
Пожароопасность
Flammable vapors when heated. Runoff from fire control water may give off poisonous gases and cause pollution. Mixtures of 9-10% phenol in air are explosive. Avoid aluminum chloride/nitrobenzene mixture, peroxodisulfuric acid, peroxomonosulfuric acid and strong oxidizing agents. Decomposes slowly on air contact. Avoid contact with strong oxidizing agents.Воспламеняемость и взрывоопасность
Phenol is a combustible solid (NFPA rating = 2). When heated, phenol produces flammable vapors that are explosive at concentrations of 3 to 10% in air. Carbon dioxide or dry chemical extinguishers should be used to fight phenol fires.Фармацевтические приложения
Phenol is used mainly as an antimicrobial preservative in parenteral pharmaceutical products. It has also been used in topical pharmaceutical formulations and cosmetics;Phenol is widely used as an antiseptic, disinfectant, and therapeutic agent, although it should not be used to preserve preparations that are to be freeze-dried.
Промышленное использование
Phenol is the simplest member of a class oforganic compounds possessing a hydroxylgroup attached to a benzene ring or to a morecomplex aromatic ring system.Also known as carbolic acid or monohydroxybenzene,phenol is a colorless to whitecrystalline material of sweet odor, having thecomposition C6H5OH, obtained from the distillationof coal tar and as a by-product ofcoke ovens.
Phenol has broad biocidal properties, anddilute aqueous solutions have long been usedas an antiseptic. At higher concentrations itcauses severe skin burns; it is a violent systemicpoison. It is a valuable chemical raw materialfor the production of plastics, dyes, pharmaceuticals,syntans, and other products.
Phenol is one of the most versatile industrialorganic chemicals. It is the starting point formany diverse products used in the home andindustry. A partial list includes nylon, epoxyresins, surface active agents, synthetic detergents,plasticizers, antioxidants, lube oil additives,phenolic resins (with formaldehyde, furfural,and so on), cyclohexanol, adipic acid,polyurethanes, aspirin, dyes, wood preservatives,herbicides, drugs, fungicides, gasolineadditives, inhibitors, explosives, and pesticides.
Безопасность
Phenol is highly corrosive and toxic, the main effects being on the central nervous system. The lethal human oral dose is estimated to be 1 g for an adult.Phenol is absorbed from the gastrointestinal tract, skin, and mucous membranes, and is metabolized to phenylglucuronide and phenyl sulfate, which are excreted in the urine.
Although there are a number of reports describing the toxic effects of phenol, these largely concern instances of accidental poisoning or adverse reactions during its use as a therapeutic agent.Adverse reactions associated with phenol used as a preservative are less likely owing to the smaller quantities that are used; however, it has been suggested that the body burden of phenol should not exceed 50 mg in a 10-hour period.This amount could be exceeded following administration of large volumes of phenolpreserved medicines.
LD50 (mouse, IV): 0.11 g/kg
LD50 (mouse, oral): 0.3 g/kg
LD50 (rabbit, skin): 0.85 g/kg
LD50 (rat, skin): 0.67 g/kg
LD50 (rat, oral): 0.32 g/kg
LD50 (rat, SC): 0.46 g/kg
Возможный контакт
Phenol is used as a pharmaceutical, in the production of fertilizer; coke, illuminating gas; lampblack, paints, paint removers; rubber, asbestos goods; wood preservatives; synthetic resins; textiles, drugs, pharmaceutical preparations; perfumes, bakelite, and other plastics (phenolformaldehyde resins); polymer intermediates (caprolactam, bisphenol-A and adipic acid). Phenol also finds wide use as a disinfectant and veterinary drug.Канцерогенность
Phenol had been investigated for carcinogenicity in animals by the oral and dermal routes. IARC and IRIS determined that animal human evidence for carcinogenicity was inadequate.хранилище
When exposed to air and light, phenol turns a red or brown color, the color being influenced by the presence of metallic impurities. Oxidizing agents also hasten the color change. Aqueous solutions of phenol are stable. Oily solutions for injection may be sterilized in hermetically sealed containers by dry heat. The bulk material should be stored in a well-closed, light-resistant container at a temperature not exceeding 15°C.Перевозки
UN1671 Phenol, solid, Hazard Class: 6.1; Labels: 6.1-Poisonous materials. UN2312 Molten phenol, Hazard Class: 6.1; Labels: 6.1-Poisonous materials. UN2821 Phenol solutions, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.Несовместимости
Phenol, available in solid or liquid form, is colorless to light pink and has a sweet aromatic odor. It is stable under normal conditions of storage and use. The liquid and vapor are combustible. Phenol is incompatible with strong oxidizing agents, calcium hypochlorite, halogens, halogenated compounds, aluminum chloride, and nitrobenzene. Hot phenol can attack aluminum, lead, magnesium and zinc. It can react exothermally with peroxymonosulfuric acid, sodium nitrate, 1,3-butadiene and boron trifluoride diethyl ether. When phenol is heated to decomposition (ca. 715 °C), decomposition products include carbon monoxide and carbon dioxide.https://www.cdc.gov/niosh/npg/npgd0493.html
http://www51.honeywell.com/sm/common/documents/Public_Risk_Summary_-_GPS0075_Phenol_Dec_2012.pdf
Утилизация отходов
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. Incineration.Меры предосторожности
Acute poisoning of phenol by ingestion, inhalation or skin contact may lead to death. Phenol is readily absorbed through the skin. It is highly toxic by inhalation. It is corrosive and causes burns and severe irritation effects. During use and handling of phenol, occupational workers should be very careful. Workers should use protective clothing, rubber boots, and goggles to protect the eyes from vapors and spillage.Регуляторный статус
Included in the FDA Inactive Ingredients Database (injections). Included in medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.Фенол запасные части и сырье
сырьё
1of4
запасной предмет
- 2-МЕТИЛ-4-ХЛОРОФЕНОКСИУКСУСНАЯ КИСЛОТА
- 3-ХЛОРО-4-ФЕНОКСИАНИЛИН
- SODIUM PHENOLATE TRIHYDRATE
- Tetrabromophenol Синий
- Бисакодил
- 3-феноксипропионова кислота
- 2,4,6-Три-трет-бутилфенол
- Dispersing agent S
- BISPHENOL A DIMETHACRYLATE
- 4-нитрофенил фениловый эфир
- Три(4-изопропилфенил)фосфат
- Antioxidant 1076
- 2,4-ди-трет-бутилфенол
- Antioxidant 168
- 3-PHENOXYANILINE
- 4'-Phenoxyacetophenone
- Пентахлорфенолят натрия
- High temperature levelling agent BOF
- P-гидроксикоричная кислота
- 6-ФЕНОКСИНИКОТИНОТРИЛ
- Roxatidine acetate
- Fenpropathrin
- ALKYLPHENOL DISULFIDE
- Sulphur Indanthrene Blue RNX
- 3-AMINO-1-PHENOXY-2-PROPANOL HYDROCHLORIDE
1of8
Фенол поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия | |
|---|---|---|---|---|---|
| +8613325218432 | China | 57 | 58 | ||
| +86-18678179670 +86-18615116763 |
China | 101 | 58 | ||
| +86-85511178; +86-85511178; |
China | 35425 | 58 | ||
| +8619931165850 | China | 1000 | 58 | ||
| +86-17736087130 +86-18633844644 |
China | 994 | 58 | ||
| +8615531157085 | China | 8804 | 58 | ||
| +862156762820 +86-13564624040 |
China | 7707 | 58 | ||
| +86-13131129325 | China | 5887 | 58 | ||
| +8615530197691 | China | 105 | 58 | ||
| +86-(0)57185586718 +86-13336195806 |
China | 29792 | 60 |