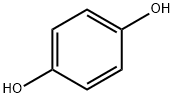
Гидрохинон
- английское имяHydroquinone
- CAS №123-31-9
- CBNumberCB6717931
- ФормулаC6H6O2
- мольный вес110.11
- EINECS204-617-8
- номер MDLMFCD00002339
- файл Mol123-31-9.mol
| Температура плавления | 172-175 °C(lit.) |
| Температура кипения | 285 °C(lit.) |
| плотность | 1.32 |
| Плотность накопления | 600kg/m3 |
| плотность пара | 3.81 (vs air) |
| давление пара | 1 mm Hg ( 132 °C) |
| показатель преломления | 1.6320 |
| Fp | 165 °C |
| температура хранения | Store below +30°C. |
| растворимость | H2O: 50 mg/mL, clear |
| пка | 10.35(at 20℃) |
| форма | Needle-Like Crystals or Crystalline Powder |
| цвет | White to off-white |
| Запах | odorless |
| Биологические источники | synthetic |
| Растворимость в воде | 70 g/L (20 ºC) |
| Чувствительный | Air & Light Sensitive |
| Мерк | 14,4808 |
| БРН | 605970 |
| констант закона Генри | (x 10-9 atm?m3/mol): <2.07 at 20 °C (approximate - calculated from water solubility and vapor pressure) |
| Пределы воздействия | NIOSH REL: 15-min ceiling 2, IDLH 50; OSHA PEL: TWA 2; ACGIH TLV: TWA 2 (adopted). |
| Стабильность | Stable. Combustible. Incompatible with strong oxidizing agents, strong bases, oxygen, ferric salts. Light and air-sensitive. Discolours in air. |
| ИнЧИКей | QIGBRXMKCJKVMJ-UHFFFAOYSA-N |
| LogP | 0.59 at 20℃ |
| Непрямые добавки, используемые в веществах, контактирующих с пищевыми продуктами | HYDROQUINONE |
| FDA 21 CFR | 175.105 |
| Справочник по базе данных CAS | 123-31-9(CAS DataBase Reference) |
| Рейтинг продуктов питания EWG | 6-9 |
| FDA UNII | XV74C1N1AE |
| Код УВД | D11AX11 |
| МАИР | 3 (Vol. 15, Sup 7, 71) 1999 |
| Справочник по химии NIST | Hydroquinone(123-31-9) |
| Система регистрации веществ EPA | Hydroquinone (123-31-9) |
| UNSPSC Code | 41116107 |
| NACRES | NA.77 |
| Коды опасности | Xn,N | |||||||||
| Заявления о рисках | 22-40-41-43-50-68-R68-R50-R43-R41-R40-R22 | |||||||||
| Заявления о безопасности | 26-36/37/39-61-S61-S36/37/39-S26 | |||||||||
| РИДАДР | 2662 | |||||||||
| OEL | Ceiling: 2 mg/m3 [15-minute] | |||||||||
| WGK Германия | 3 | |||||||||
| RTECS | MX3500000 | |||||||||
| Температура самовоспламенения | 930 °F | |||||||||
| TSCA | Yes | |||||||||
| Класс опасности | 9 | |||||||||
| Группа упаковки | III | |||||||||
| кода HS | 29072210 | |||||||||
| Банк данных об опасных веществах | 123-31-9(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 orally in rats: 320 mg/kg (Woodard) | |||||||||
| ИДЛА | 50 mg/m3 | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)




-
сигнальный язык
опасность
-
вредная бумага
H302:Вредно при проглатывании.
H318:При попадании в глаза вызывает необратимые последствия.
H341:Предполагается, что данное вещество вызывает генетические дефекты.
H351:Предполагается, что данное вещество вызывает раковые заболевания.
H410:Чрезвычайно токсично для водных организмов с долгосрочными последствиями.
-
оператор предупредительных мер
P202:Перед использованием ознакомиться с инструкциями по технике безопасности.
P273:Избегать попадания в окружающую среду.
P280:Использовать перчатки/ средства защиты глаз/ лица.
P301+P312:ПРИ ПРОГЛАТЫВАНИИ: Обратиться за медицинской помощью при плохом самочувствии.
P305+P351+P338:ПРИ ПОПАДАНИИ В ГЛАЗА: Осторожно промыть глаза водой в течение нескольких минут. Снять контактные линзы, если Вы ими пользуетесь и если это легко сделать. Продолжить промывание глаз.
P308+P313:ПРИ подозрении на возможность воздействия обратиться за медицинской помощью.
Гидрохинон химические свойства, назначение, производство
Описание
Hydroquinone (HQ) is produced by the oxidation of aniline or phenol, by the reduction of quinone, or from a reaction of acetylene and carbon monoxide. Hydroquinone occurs naturally as a glucose ether, also known as arbutin, in the leaves of many plants and in fruits, as well as one of the agents used in the defense mechanism of the bombardier beetle, family Carabidae.Химические свойства
Hydroquinone, a colorless, hexagonal prism, has been reported to be a good antimitotic and tumor-inhibiting agent. It is a reducing agent used in a photographic developer, which polymerizes in the presence of oxidizing agents. In the manufacturing industry it may occur include bacteriostatic agent, drug, fur processing, motor fuel, paint, organic chemicals, plastics, stone coating, and styrene monomers.Физические свойства
Colorless to pale brown, odorless, hexagonal crystalsИспользование
hydroquinone is a pigment-lightening agent used in bleaching creams. Hydroquinone combines with oxygen very rapidly and becomes brown when exposed to air. Although it occurs naturally, the synthetic version is the one commonly used in cosmetics. Application to the skin may cause allergic reaction and increase skin sun sensitivity. Hydroquinone is potentially carcinogenic and is associated with causing ochronosis, a discoloration of the skin. The u.S. FDA has banned hydroquinone from oTC cosmetic formulations, but allows 4 percent in prescription products. Its use in cosmetics is prohibited in some european countries and in Australia.Определение
ChEBI: A benzenediol comprising benzene core carrying two hydroxy substituents para to each other.Методы производства
There are three current manufacturing processes for HQ: oxidative cleavage of diisopropylbenzene, oxidation of aniline, and hydroxylation of phenol.Diisopropylbenzene is air oxidized to the intermediate diisopropylbenzene bishydroperoxide. This hydroperoxide is purified by extraction and reacted further to form hydroquinone. The purified product is isolated by filtration and packaged. The process can be almost entirely closed, continuous, computer-controlled, and monitored.
HQcan also be prepared by oxidizing aniline to quinone in the presence of manganese dioxide and sulfuric acid. p-Benzoquinone is then reduced to HQ using iron oxide. The resulting hydroquinone is crystallized and dried. The process occurs in a closed system.
HQis also manufactured by hydroxylation of phenol using hydrogen peroxide as a hydroxylation agent. The reaction is catalyzed by strong mineral acids or ferrous or cobalt salts.
Показания
Hydroquinone interferes with the production of the pigment melanin by epidermal melanocytes through at least two mechanisms: it competitively inhibits tyrosinase, one of the principal enzymes responsible for converting tyrosine to melanin, and it selectively damages melanocytes and melanosomes (the organelles within which melanin is stored).Всемирная организация здравоохранения(ВОЗ)
Hydroquinone was introduced in 1965 as a topical depigmenting agent for hyperpigmentation. At high concentrations hydroquinone is corrosive and in most countries has been restricted to the level of approximately 2% and limited to the period of less than 2 months. Additional consideration for restrictive action is that animal experiments have also demonstrated carcinogenic and mutagenic potential of hydroquinone.Общее описание
Light colored crystals or solutions. May irritate the skin, eyes and mucous membranes. Mildly toxic by ingestion or skin absorption.Реакции воздуха и воды
Darkens on exposure to air and light. Miscible in water. Solutions become brown in air due to oxidation. Oxidation is very rapid in the presence of alkali.Профиль реактивности
Hydroquinone is a slight explosion hazard when exposed to heat. Incompatible with strong oxidizing agents. Also incompatible with bases. Hydroquinone reacts with oxygen and sodium hydroxide. Reacts with ferric salts . Hot and/or concentrated NaOH can cause Hydroquinone to decompose exothermically at elevated temperature. (NFPA Pub. 491M, 1975, 385)Опасность
Toxic by ingestion and inhalation, irritant. Questionable carcinogen.Угроза здоровью
Hydroquinone is very toxic; the probable oral lethal dose for humans is 50-500 mg/kg, or between 1 teaspoon and 1 ounce for a 150 lb. person. It is irritating but not corrosive. Fatal human doses have ranged from 5-12 grams, but 300-500 mg have been ingested daily for 3-5 months without ill effects. Death is apparently initiated by respiratory failure or anoxia.Пожароопасность
Dust cloud may explode if ignited in an enclosed area. Hydroquinone can react with oxidizing materials and is rapidly oxidized in the presence of alkaline materials. Oxidizes in air.Контактные аллергены
Hydroquinone is used in photography developers (black and white, X-ray, and microfilms), in plastics, in hair dyes as an antioxidant and hair colorant. Hydroquinone is found in many skin bleaching creams.Клиническое использование
Hydroquinone is applied topically to treat disorders characterized by excessive melanin in the epidermis, such as melasma. In the United States, nonprescription skin-lightening products contain hydroquinone at concentrations of 2% or less; higher concentrations are available by prescription.Побочные эффекты
The incidence of adverse effects with hydroquinone increases in proportion to its concentration. A relatively common side effect is local irritation, which may actually exacerbate the discoloration of the skin being treated. Allergic contact dermatitis occurs less commonly. A rare but more serious complication is exogenous ochronosis, in which a yellow-brown pigment deposited in the dermis results in blue-black pigmentation of the skin that may be permanent.Канцерогенность
No case reports of cancer associated with HQ exposure have been published.Методы очистки
Crystallise quinol from acetone, *benzene, EtOH, EtOH/*benzene, water or acetonitrile (25g in 30mL), preferably under nitrogen. Dry it under vacuum. [Wolfenden et al. J Am Chem Soc 109 463 1987, Beilstein 6 H 836, 6 IV 5712.]Гидрохинон запасные части и сырье
сырьё
1of3
запасной предмет
- ХЛОРАЛЬГИДРАТ
- трет-Бутилгидрохинон
- 4-Хлор-2 ,5-диметоксианилина
- этил-3,3-диметилпент-4-ен-1-оат
- Fluazifop-P-butyl
- Hydroxypropyl acrylate
- 1-(2,5-Dimethoxyphenyl)-2-oximino-1-propanone
- 7-HYDROXYISOFLAVONE
- 4-Хлор-8-(трифторметил) хинолин
- Бутилгидроксианизол
- Энбукрилат
- 2-амино-1- (2,5-диметоксифенил) -1-пропанон
- Пиреноксин
- X-PHOS
- 3-METHYL-2,5-DIHYDROTHIOPHENE-1,1-DIOXIDE
- 2-[N-(2-Cyanoethyl)-4-[(2,6-dichloro-4-nitrophenyl)azo]anilino]ethyl acetate
- ammonium manganous sulfate
- 1,2,4-триметоксибензол
- Хлор(1,5-циклооктадиен)иридий(I) димер
- 3-Sulfolene
- Fluazifop-butyl
- 2-ГИДРОКСИЭТИЛМЕТАКРИЛАТ
- methyl(±)cis,trans-2,2-dimethyl-3-(2-methyl-1-propenyl cyclopropane carboxylate)
- N-NONYL ACRYLATE
- Быстрый синий BB
- 2,3-диметил-1,3-бутадиен
- 2,5-дигидроксибензойной кислоты
- 4-[(6-Chloro-1,3-benzoxazol-2-yl)oxy]phenol
- 4-Phenoxyphenol
- 1,4-ДИМЕТОКСИБЕНЗОЛ
1of8
Гидрохинон поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия | |
|---|---|---|---|---|---|
| 0512-52678575 13776233393 |
China | 212 | 58 | ||
| +8615531157085 | China | 8804 | 58 | ||
| +86-0551-65418671 +8618949823763 |
China | 34563 | 58 | ||
| +8617320528784 | China | 41 | 58 | ||
| +8619931165850 | China | 1000 | 58 | ||
| +8615350571055 | China | 6087 | 58 | ||
| +86-13131129325 | China | 5887 | 58 | ||
| +8613288715578 | China | 1174 | 58 | ||
| +86-(0)57185586718 +86-13336195806 |
China | 29792 | 60 | ||
| +86-0371-55170693 +86-19937530512 |
China | 21632 | 55 |
Гидрохинон Обзор)
1of4