The synthesis method of Malonic acid
Introduction
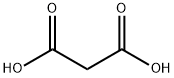
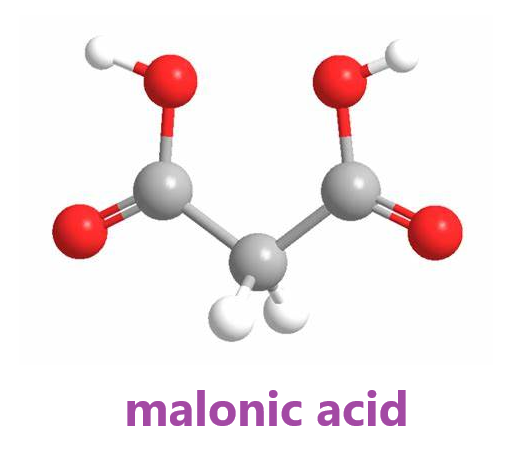
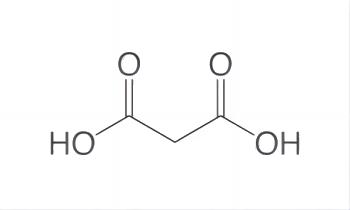
Malonic acid (IUPAC systematic name: propanedioic acid) is a dicarboxylic acid with structure CH2(COOH)2. Dicarboxylic acids are organic compounds containing two functional groups of carboxylic acid. Dicarboxylic acids generally show the same chemical behaviour and reactivity as monocarboxylic acids. The ionized form of malonic acid and its esters and salts are known as malonates. For example, diethyl malonate is malonic acid's diethyl ester. The name originates from the Greek word μᾶλον (malon), meaning 'apple'.
Natural source
Malonic acid is found in small amounts in sugar beet and green wheat, formed by oxidative degradation of malic acid. Malonic acid has been identified in celery, parsnip, green pepper, tangerine, beet, oats, tabasco, chili, orange, grapefruit, lime, melon, cantaloupe, sunflower, barley, prickly pear, Indian fig, ginseng, black bean, kidney bean, green bean, peach, corn, Scotch pine, alfalfa, banana, tobacco plant, and juniper.
Synthesis
Malonic acid is widely used in manufacturing processes, such as the petrochemical, pharmaceutical, and cosmetic industries. Traditionally, malonic acid is produced from fossil resources via petrochemical processes. The byproduct sodium cyanide is dangerous and unfriendly to the environment. With increasing concerns about energy and environmental problems, producing malonic acid by microbial fermentation via bioconversion of renewable feedstock has generated considerable interest worldwide.
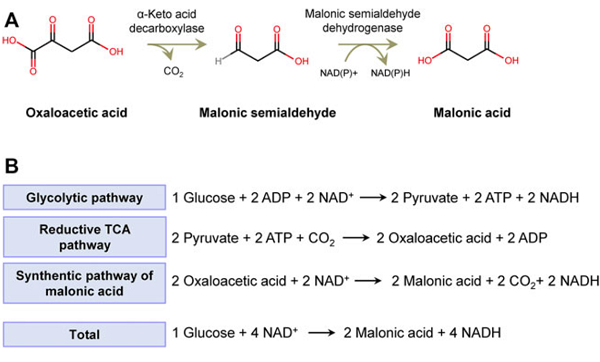
Currently, two non-natural metabolic pathways have been sought and introduced into microbes, using malonic semialdehyde (MSA) or malonyl-CoA as the precursor. The first uses MSA from the deamination reaction of β-alanine as the precursor. In Escherichia coli, β-alanine pyruvate transaminase from Pseudomonas aeruginosa was overexpressed to convert β-alanine to produce MSA. Then, succinate semialdehyde dehydrogenase encoded by E. coli yneI was used to catalyze the reaction of MSA to malic acid, and the titer of malonic acid reached 3.60 g/L by fed-batch fermentation. Another pathway depends on the fatty acid synthesis pathway, using malonyl-CoA as the precursor. Matthew et al. reported that acyl-CoA hydrolase encoded by ehd3 from Saccharomyces cerevisiae can hydrolyze malonyl-CoA to malonic acid. During overexpression of EHD3 in S. cerevisiae, Pichia kudriavzevii, and E. coli, malonic acid in the cultures was detectable. After the improvement of the binding affinity of acyl-CoA hydrolase to malonyl-CoA by protein engineering, the production of malonic acid was increased to 0.0823 g/L by engineered E. coli. Both synthetic pathways of malonic acid require several enzymes to convert glucose to target acid and are complex, which might lead to a low titer of malonic acid.
Hence, Gu et al. designed a novel artificial synthetic pathway of malonic acid, in which oxaloacetate, an intermediate of cytoplasmic reductive tricarboxylic acid (TCA) pathway, is converted to malonic semialdehyde and then to malonic acid, sequentially catalyzed by a-keto decarboxylase and malonic semialdehyde dehydrogenase. After the systematic screening, researchers discovered the enzyme oxaloacetate decarboxylase Mdc, catalyzing the first step of the artificially designed pathway in vitro. Then, this synthetic pathway was functionally constructed in cellulolytic thermophilic fungus Myceliophthora thermophila. After enhancement of glucose uptake, the titer of malonic acid achieved 42.5 mg/L.
References:
[1] SHUYING GU. Designing and Constructing a Novel Artificial Pathway for Malonic Acid Production Biologically.[J]. ACS Applied Energy Materials, 2022. DOI:10.3389/fbioe.2021.820507.You may like
Related articles And Qustion
See also
Lastest Price from Malonic acid manufacturers

US $120.00/kg2025-08-02
- CAS:
- Min. Order:
- 1kg
- Purity:
- 99
- Supply Ability:
- 60 tons

US $1.00/kg2025-04-21
- CAS:
- 141-82-2
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt