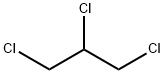
1,2,3-Trichloropropane is a chemical compound existing as an intermediate in certain pesticides. It is used as the intermediate for the manufacturing of chemicals such as epoxy resins, polysulfone liquid polymers, the synthesis of hexafluoropropylene, and as a cross-linking agent in the synthesis of polysulfides. It has also been used as the solvent for paint and varnish removal, a cleaning and degreasing agent, and a maintenance solvent. It has also been used as a soil fumigant. In addition, it is also a “branching” or curing agent in polysulfide polymers, which is used as the sealants for aircraft fuel tanks and bodies. However, it should be noted that it is a persistent groundwater pollutant and a potential human carcinogen.
1,2,3-Trichloropropane (hereinafter referred to as TCP) is a chlorinated hydrocarbon that was historically used as an industrial solvent and a degreasing agent. Currently, however, TCP is utilized as an intermediate in the production of polymer cross-linking agents, pesticides, and glycerol. In its pure form, TCP is a colorless to yellow liquid with limited solubility in water, a strong chloroform-like odor, moderate volatility, and high flammability.
Produced in large quantities as an epichlorohydrin production byproduct, TCP is a synthetic compound that does not occur naturally in the environment. In the agrochemical industry, TCP is formed via the manufacture of dichloropropene-derived nematicides (pesticides used to kill parasitic nematodes), and it is also present as an impurity in these soil fumigants. As a result, application of these products has produced significant atmosphere, soil, and groundwater contamination, which in turn can induce various health problems in wildlife and humans. The toxicological effects of TCP depend on dose and duration, but can range from kidney and liver damage to tumors and cancers.
The EPA has established drinking water health advisories for 1,2,3-Trichloropropane, which are drinking water specific risk level concentrations for cancer (10-4 cancer risk) and concentrations of drinking water contaminants at which noncancer adverse health effects are not anticipated to occur over specific exposure durations. EPA established a 1-day health advisory of 0.6 milligrams per liter (mg/L) and a 10-day health advisory of 0.6 mg/L for TCP in drinking water for a 10 kilogram (kg) child (EPA 2012a).
- Samin, G, and D. B. Janssen. "Transformation and biodegradation of 1,2,3-trichloropropane (TCP). " Environmental Science & Pollution Research 19.8(2012): 3067-3078.
- Liu, P., et al. "Successful therapy with hemoperfusion and plasma exchange in acute 1,2,3-trichloropropane poisoning." Human & Experimental Toxicology 31.5(2012): 523-527.
1,2,3-Trichloropropane is a synthetic chemical that is also known as allyl trichloride, glycerol trichlorohydrin, and trichlorohydrin. It is a colourless, heavy liquid with a sweet but strong chloroform-like odour and is combustible. It is slightly soluble in water but soluble in chloroform, diethyl ether, and ethanol. On contact with heat/ fire, It releases off irritating or toxic fumes (or gases). It evaporates very quickly and small amounts dissolve in water. It is mainly used to make other chemicals. 1,2,3-Trichloropropane was used in the past mainly as a solvent and extractive agent, including as a paint and varnish remover and as a cleaning and degreasing agent. It is now used mainly as a chemical intermediate, for example, in the production of polysulphone liquid polymers, dichloropropene and hexafluoropropylene and as a cross-linking agent in the synthesis of polysulphides.
1,2,3-Trichloropropane is a colorless liquid with a strong acid odor. slightly soluble in water; dissolves oils, fats, waxes, chlorinated rubber, and numerous resins. autoign temp 580F (304°C). Combustible.
Clear, colorless liquid with a strong, chloroform-like odor
Intermediate in the manufacture of
pesticides and polysulfide rubbers; formerly
used as a solvent and extractive agent.
Paint and varnish remover, solvent, degreasing
agent.
1,2,3-Trichloropropane is used as a solventand as an intermediate in organic synthesis.
ChEBI: 1,2,3-Trichloropropane is an organochlorine compound.
Colorless liquid with a strong acid odor. Denser than water and slightly soluble in water. Hence sinks in water.
1,2,3-Trichloropropane is sensitive to prolonged exposure to light. Sensitive to heat. May react with active metals, strong caustics and oxidizing agents. Attacks some plastics, rubber and some coatings .
Inhalation of vapor causes anesthesia, dizziness, and nausea. Vapor is highly irritating by inhalation routes and moderately irritating by dermal routes. Exposure of eyes to vapor may result in slight, transient injury to the cornea.
Inhalation of its vapors can produce depres sion of the central nervous system, which canprogress to narcosis and convulsion as theconcentration increases. A 30-minute expo sure to a 5000-ppm concentration causedconvulsions in rats. Acute as well as chronicexposure to high concentrations can causeliver damage. 1,2,3-Trichloropropane is moretoxic than its 1,1,1-isomer. Acute oral tox icity is moderate, with LD50 values rang ing between 300 and 550 mg/kg in differentspecies of experimental animals. The liquidis a strong irritant to the eyes.
Combustible liquid; flash point (closed cup)
73°C (164°F), (open cup) 82°C (180°F).
Vapors of 1,2,3-trichloropropane form explo sive mixtures with air, with LEL and UEL
values of 3.2% and 12.6% by volume in air,
respectively. The compound reacts vigorously
with alkali metals, powdered magnesium, or
aluminum; caustic alkalies; and oxidizers.
Flammability and Explosibility
Non flammable
Confirmed carcinogen.
Poison by ingestion. Moderately toxic by
inhalation and skin contact. Experimental
reproductive effects. A skin and severe eye
irritant. Mutation data reported. Moderately
flammable by heat, flames (sparks), or
powerful oxidizers. See also ALLYL
COMPOUNDS and CHLORINATED
HYDROCARBONS, ALIPHATIC. When
heated to decomposition it yields hghly
toxic Cl-. To fight fre, use water (as a
blanket), spray, mist, dry chemical.
Trichloropropane dissolves oils, fats,
waxes, chlorinated rubber; and numerous resins; it is used
as a paint and varnish remover; a solvent; and a degreasing
agent.
1,2,3-Trichloropropane is reasonably anticipated to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in experimental animals.
Chemical/Physical. The hydrolysis rate constant for 1,2,3-trichloropropane at pH 7 and 25 °C
was determined to be 1.8 x 10-6/h, resulting in a half-life of 43.9 yr (Ellington et al., 1988). The
hydrolysis half-lives decrease at varying pHs and temperature. At 87 °C, the hydrolysis half-lives
at pH values of 3.07, 7.12, and 9.71 were 21.1, 11.6, and 0.03 d, respectively (Ellington et al.,
1986). By analogy to 1,2-dibromo-2-chloropropane, the following hydrolysis products would be
formed: 2,3-dichloro-1-propanol, 2,3-dichloropropene, epichlorohydrin, 1-chloro-2,3-
dihydroxypropane, glycerol, 1-hydroxy-2,3-propylene oxide, 2-chloro-3-hydroxypropene, and
HCl (Kollig, 1993).
The volatilization half-life of 1,2,3-trichloropropane (1 mg/L) from water at 25 °C using a
shallow-pitch propeller stirrer at 200 rpm at an average depth of 6.5 cm was 56.1 min (Dilling,
1977).
UN2810 Toxic liquids, organic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical
Name Required.
It is currently believed that TCP itself has very low activity,
while its metabolites mainly cause toxicity, carcinogenicity, and
other effects. The liver can metabolize TCP by cytochrome P450
enzymes to give reactive intermediates, which bind to glutathione
or other protein for excretion. The reactive intermediates
can bind to DNA, proteins, and other molecules and cause
hepatocellular damage, gene mutation, and organ dysfunction.
Activation of the molecule may occur by reaction with glutathione.
The metabolites (e.g., dichloroacetone) accumulated
through glutathione depletion and consequently covalent
binding to DNA and other macromolecules such as microsomal
proteins.
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions.
Keep away from alkaline materials, strong bases,
strong acids, oxoacids, epoxides. Violent decomposition
with chemically active metals; strong bases. Keep away
from chlorinated rubber, resins and waxes; and sunlight.
The current initial threshold screening level (ITSL) for 1,2,3-trichloropropane is 0.3 μg/m3 based on an annual averaging time.
Incineration, preferably after
mixing with another combustible fuel. Care must be exercised
to assure complete combustion to prevent the formation
of phosgene. An acid scrubber is necessary to remove
the halo acids produced.