Yellowish brown crystalline solid. Melting point 122-123°C, vapor pressure 1.33×10-3 Pa. Insoluble in water, soluble in aromatic hydrocarbons. Stable to aqueous acid and alkali, but under high temperature conditions or sunlight UV irradiation will lose activity, 130 ℃ 6h activity decreased by 50%.
Dienochlor is used for the control of mites (Tetrunychus spp.,
Panonychus ulmi and Polyphagotarsonemus latus) on roses, Chrysanthemums
and other ornamentals.
Acaricide used for control of mites on ornamentals.
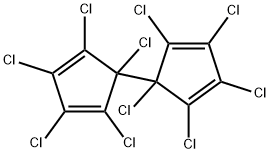
ChEBI: Dienochlor is an organochlorine compound.
Plant. On plants, dienochlor was converted by sunlight to form perchloro ketones (Quistad and Mulholland, 1983).
Chemical/Physical. Dienochlor is unstable when exposed to sunlight. When dienochlor applied as a thin lm on glass plates was exposed to sunlight, nonpolar products, a tricyclic chlorocarbon and 3 isomeric perchloro ketones were formed at yields
Dienochlor begins to decompose at 130°C (Worthing and Hance, 1991).
Dienochlor is readily degraded in sunlight to many products, only a few
of which have been identified. It is also metabolised in animals but to
unknown products. In vitro studies have shown that it interacts with thiols
(glutathione, cysteine, efc.) and proteins.
Dienochlor decomposes in simulated sunlight (DT50
1.6 min). Degradation is mainly environmental rather
than metabolic, and photochemical breakdown is rapid. It
decomposes in soils, DT50 3.1 days and DT50 2–3 days on
plants exposed to sunlight.The major degradation products of dienochlor in plants are perchloroketones. It is rapidly degraded in rats.
Dienochlor is stable in storage at 54°C for 14 days and at 42°C over
2 years. It undergoes hydrolysis with DT50 values at pH 5,7 and 9 of 184,
93 and 30.5 days, respectively, at 25 °C. It decomposes in simulated sunlight
with a DT50 of 1.6 minutes (PM).
A thin film of dienochlor on glass was readily degraded with a half-life
of <1 hour on exposure to sunlight. Four photoproducts were isolated
from a plethora of products and identified by 13C NMR spectroscopy
(Quistad and Mulholland, 1983). The photocatalysed addition of two
chlorine atoms (from donor dienochlor) afforded the tricyclic photochlorination
product 2 (up to 10% yield). Three isomeric perchloroketones
(3,4 and 5), each resulting from the net addition of one oxygen atom, were
major products, both on glass (up to 14% combined isomers) and on
cucumber and strawberry plants (up to 11% yield). Generally products 2,
3 and 4 were found in similar yields and 5 was the minor of the identified
metabolites. The photochlorination product 2, however, was not detected
on the plants. These products are illustrated in Scheme 1.