Clopidogrel was launched in the US as a potent inhibitor of platelet
aggregation for the preventive management of secondary ischemic events,
including MI, stroke and vascular deaths. Clopidogrel can be synthesized in 4
steps (including an optical resolution to the S active enantiomer) from 2-(2-
ch1orophenyl)-glycine, the key step being the cyclization to thienopyridine with
formaldehyde and acetic acid. Clopidogrel belongs to the original chemical class
of Ticlopidine, but shows fewer side effects (in particular, bone-marrowsuppressing
effects) at the dosage generally used. Like Ticlopidine, it is an
Adenosine diphosphate (ADP) antagonist acting at the purinergic P2y receptor.
In in vivo experiments with rabbits, Clopidogrel shows a maximal antiaggregant
effect at 20mg/kg po, reducing adhesion of platelets to the vascular
subendothelium ; moreover, it reduces myointimal thickening occuring after
endothelial injury of rat carotid artery. Clopigrel does not affect platelet
aggregation in vitro ; actually, its in vivo activity is highly dependent on hepatic
metabolism. The results of a CAPRIE trial (Clopidogrel versus Aspirin in patients
at risk of ischemic events) demonstrated that Clopidogrel was well tolerated and
more effective than aspirin.
Clopidogrel, sold under the brand name Plavix among others, is an antiplatelet medication used to reduce the risk of heart disease and stroke in those at high risk. Clopidogrel was discovered by Dr Fernand Eloy, it was patented in 1982, and approved for medical use in 1997. It is on the World Health Organization's List of Essential Medicines.
anthelmintic, antiparasitic, antimite
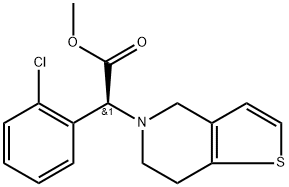
ChEBI: Clopidogrel is a thienopyridine that is 4,5,6,7-tetrahydrothieno[3,2-c]pyridine in which the hydrogen attached to the nitrogen is replaced by an o-chlorobenzyl group, the methylene hydrogen of which is replaced by a methoxycarbonyl group (the S enantiomer). A P2Y12 receptor antagonist, it is used to inhibit blood clots and prevent heart attacks. It has a role as a platelet aggregation inhibitor, an anticoagulant and a P2Y12 receptor antagonist. It is a thienopyridine, a member of monochlorobenzenes and a methyl ester. It is functionally related to a ticlopidine.
Plavix (Sanofi Aventis);Plavix, Iscover.
Clopidogrel, methyl (+)-(S)-α-(2-chlorophenyl)-6,7-dihydrothieno[3,2-c]pyridine-5(4H)-acetate sulfate (Plavix), is useful for the preventativemanagement of secondary ischemic events, including myocardialinfarction, stroke, and vascular deaths. It may beclassified as a thienopyridine because of its heterocyclicsystem. Several agents possessing this system have beenevaluated as potential antithrombotic agents. These agentshave a unique mechanism, in that they inhibit the purinergicreceptor located on platelets. Normally, nucleotides act asagonists on these receptors, which include the P2Y type.Two P2Y receptor subtypes (P2Y1 and P2Y2) found onplatelets, when stimulated by adenosine diphosphate (ADP),cause platelet aggregation.
Clopidogrel acts as an antagonistto the P2Y2 receptor. It is probably a prodrug that requiresmetabolic activation, because in vitro studies do not interferewith platelet aggregation. Although platelet aggregationis not normally seen in the first 8 to 11 days after administrationto a patient, the effect lasts for several days after thedrug therapy is discontinued. Unlike other thienopyridinescurrently used, clopidogrel does not seriously reduce thenumber of white cells in the blood, and therefore, routinemonitoring of the white blood cell count is not necessaryduring treatment.
Potentially hazardous interactions with other drugs
Antibacterials: antiplatelet effect possibly reduced by
erythromycin.
Anticoagulants: enhanced anticoagulant effect with
coumarins and phenindione; manufacturer advises to
avoid with warfarin.
Heparin: increased risk of bleeding.
Antidepressants: antiplatelet effect possibly reduced
by fluoxetine, fluvoxamine and moclobemide.
Anti-diabetics: avoid with repaglinide if possible due
to increased repaglinide exposure.
Antiepileptics: antiplatelet effect possibly reduced by
carbamazepine and oxcarbazepine.
Antifungals: antiplatelet effect possibly reduced
by fluconazole, itraconazole, ketoconazole and
voriconazole.
Antivirals: antiplatelet effect possibly reduced by
etravirine.
Statins: concentration of rosuvastatin increased,
maximum rosuvastatin dose is 20 mg.
Ulcer healing drugs: antiplatelet effect possibly
reduced by cimetidine, lansoprazole, pantoprazole
and rabeprazole; antiplatelet effect reduced by
omeprazole and esomeprazole - avoid concomitant
use if possible.
Clopidogrel is a prodrug and is extensively metabolised in
the liver, mainly to the inactive carboxylic acid derivative;
metabolism is mediated by cytochrome P450 isoenzymes
including CYP3A4 and CYP2B6, CYP1A2, CYP1A1,
and CYP2C19. The active metabolite appears to be a
thiol derivative
Clopidogrel and its metabolites are excreted in urine and
in faeces; about 50% of an oral dose is recovered from the
urine and about 46% from the faeces.