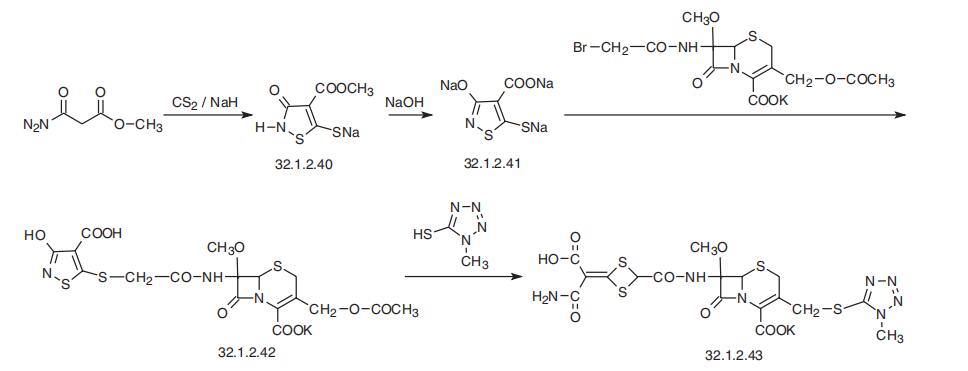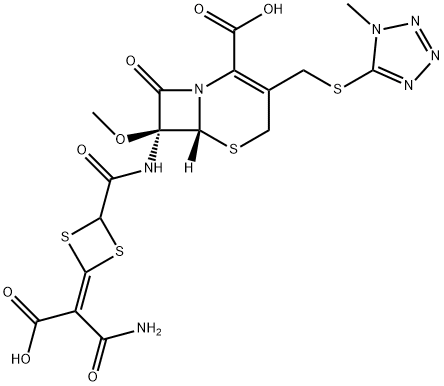
Cefotetan
- Product NameCefotetan
- CAS69712-56-7
- MFC17H17N7O8S4
- MW575.62
- EINECS274-093-3
- MOL File69712-56-7.mol
Chemical Properties
| Melting point | 173-178°C (dec.) |
| Density | 1.5157 (rough estimate) |
| refractive index | 1.7400 (estimate) |
| storage temp. | Store at 0-5°C |
| solubility | DMSO (Slightly), Methanol (Very Slightly) |
| form | Solid |
| pka | 1.69±0.41(Predicted) |
| color | White to Off-White |
| Merck | 14,1934 |
| Stability | Hygroscopic |
| InChIKey | SRZNHPXWXCNNDU-IXOPCIAXSA-N |
| SMILES | N12[C@@]([H])([C@@](NC(C3S/C(=C(\C(O)=O)/C(N)=O)/S3)=O)(OC)C1=O)SCC(CSC1N(C)N=NN=1)=C2C(O)=O |
| CAS DataBase Reference | 69712-56-7(CAS DataBase Reference) |