Description
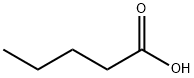
Valeric acid (also pentanoic acid) is a straight chain alkyl carboxylic acid.
Valeric acid is mainly used for the synthesis of its esters used in foods, perfume, and cosmetics. Valeric acid is used as an intermediate for applications including ester type lubricants (in aviation turbine oils, fire-resistance hydraulic fluids, and refrigerator oils), plasticizers (dipropyl heptyl phthalate), vinyl stabilizers, and specialty chemicals. It is also used as an odorant in pesticide formulations for use on crops.
References
[1] http://www.chemicalland21.com
[2] Patent CA 2496307 C: High viscosity synthetic ester lubricant base stock
[3] https://www.icis.com
[4] Christopher G. Morris (1992) Academic Press Dictionary of Science and Technology
Description
Valeric acid, or pentanoic acid, is a straight - chain alkyl carboxylic acid with the chemical formula C
5H
10O
2. Like other lowmolecular- weight carboxylic acids, it has a very unpleasant odor. It is found naturally in the perennial flowering plant valerian (Valeriana officinalis), from which it gets its name. Its primary use is in the synthesis of its esters. Volatile esters of valeric acid tend to have pleasant odors and are used in perfumes and cosmetics. Ethyl valerate and pentyl valerate are used as food additives because of their fruity flavors.
Valeric acid appears similar in structure to GHB and the neurotransmitter GABA in that it is a short-chain carboxylic acid, although it lacks the alcohol and amine functional groups that contribute to the biological activities of GHB and GABA, respectively. It differs from valproic acid simply by lacking a 3- carbon side - chain.
Chemical Properties
Colorless liquid; penetrating odor and
taste. Soluble in water,
alcohol and ether. Undergoes reactions typical of
normal monobasic organic acids. Combustible.
Chemical Properties
Valeric acid has an unpleasant odor and flavor, similar to butyric acid. May consist of one or a mixture of isomers of
n-pentanoic acid and/or 2- or 3-methyl-butanoic acid.
Occurrence
The acid is not too common in nature; reported (as the corresponding ester) found in the essential oil of Boronia
anemonifolia, in pineapple fruits and in other plants; also identified as acid or the corresponding ester in the essential oil of lemon petitgrain.
Also reported found in apple, apple juice, banana, orange juice, bilberry, cranberry, strawberry, raspberry, papaya, grapes, celery,
onion, baked potato, tomato, corn mint oil, breads, cheeses, milk, yogurt, butter, cheddar cheese, lean and fatty fish, fish oil, cooked
meats, hop oil, beer, rum, whiskies, grape wines, cocoa, tea, roasted filberts, peanuts and pecans, honey, soybeans, coconut meat and
milk, cloudberry, passion fruit, starfruit, trassi, mango, jackfruit, licorice, calamus, sake, buckwheat, watercress, laurel, peated malt,
wort, kiwifruit, loquat, Bourbon vanilla, shrimps, oyster, scallop, cape gooseberry, sea buckthorn, Chinese quince and maté.
Uses
valeric acid is obtained from valerian extract, which is considered skin conditioning.
Uses
Sex attractant of the sugar beet wireworm, Limonius californicus.1
Uses
Valeric acid, is used as a sex attractant of the sugar beet wireworm, Limonius californicus. It is used predominantly as an intermediate in the manufacture of flavors and perfumes, ester type lubricants, plasticizers and vinyl stabilizesrs. It is a food additive used as a synthetic flavoring substance dan adjuvant. It is an inert ingredient in controlling pest.
Preparation
By oxidation of n-amyl alcohol or, together with other isomers, by distillation of valerian roots; also by reacting butyl
bromide and sodium cyanide with subsequent saponification of the formed butyl nitrile.
Definition
ChEBI: A straight-chain saturated fatty acid containing five carbon atoms.
Aroma threshold values
Detection: 940 ppb to 3 ppm
Taste threshold values
Taste characteristics at 100 ppm: acidic, dairy-like with milky and cheese nuances.
General Description
A colorless liquid with a penetrating unpleasant odor. Density 0.94 g / cm3. Freezing point -93.2°F (-34°C). Boiling point 365.7°F (185.4°C). Flash point 192°F (88.9°F). Corrosive to metals and tissue.
Air & Water Reactions
Water soluble.
Reactivity Profile
Valeric acid is a carboxylic acid. Exothermically neutralizes bases, both organic and inorganic, producing water and a salt. Can react with active metals to form gaseous hydrogen and a metal salt. Reacts with cyanide salts to generate gaseous hydrogen cyanide. Flammable and/or toxic gases and heat are generated by reaction with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. Reacts with sulfites, nitrites, thiosulfates and dithionites to generate flammable and/or toxic gases and heat. Reacts with carbonates and bicarbonates to generate a harmless gas (carbon dioxide) but still heat. Can be oxidized by strong oxidizing agents and reduced by strong reducing agents. These reactions generate heat. May initiate polymerization reactions. May catalyze (increase the rate of) chemical reactions.
Health Hazard
Corrosive. Very destructive to tissues of the mucous membranes, upper respiratory tract, eyes, and skin. Symptoms may include burning sensation, coughing, wheezing, laryngitis, shortness of breath, nausea and vomiting. Extremely destructive to skin. May be absorbed through the skin.
Fire Hazard
Special Hazards of Combustion Products: Irritating vapors and toxic gases, such as carbon dioxide and carbon monoxide, may be formed when involved in fire.
Flammability and Explosibility
Non flammable
Safety Profile
Moderately toxic by
ingestion, intravenous, and subcutaneous
routes. Mildly toxic by inhalation. A
corrosive irritant to skin, eyes, and mucous
membranes. Combustible liquid. When
heated to decomposition it emits acrid
smoke and irritating fumes. Used in
perfumes.
Purification Methods
Water is removed from the acid by distillation using a Vigreux column (p 11), until the boiling point reaches 183o. A few crystals of KMnO4 are added, and after refluxing, the distillation is continued. [Andrews & Keefer J Am Chem Soc 83 3708 1961, Beilstein 2 H 299, 2 I 130, 2 II 263, 2 III 663, 2 IV 868.]