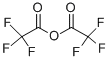
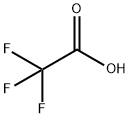
Description
Trifluoroacetic acid anhydride (TFAA), a perfluoroacylated acylation reagent, forms stable, volatile derivatives with alcohols, amines, and phenols.
Chemical Properties
Clear, colorless liquid
Uses
Trifluoroacetic anhydride is used for the introduction of trifluoroacetyl group in organic synthesis. It is involved in the preparation of N- and O-trifluoroacetyl derivatives of a wide range of biologically active compounds for gas chromatography analysis. It plays an important role as desiccant for trifluoroacetic acid. It is also used in the oxidation of aldehydes to acids, esters, amides as well as in the protection of alcohols and amines. In addition, it is used as analytical reagents, solvents and dehydration condensing agent. It serves as an intermediate of fluorine fine chemicals, pharmaceuticals and agrochemicals.
Uses
Used as analytical reagents, solvents, catalysts, dehydration condensing agent, protection agent of trifluoroacetylation of carboxyl and amino; used as Intermediate of fluorine fine chemicals, pharmaceutical, agrochemical.
Preparation
Trifluoroacetic anhydride is prepared by reaction of trifluoroacetyl chloride with an alkali metal or alkaline-earth metal salt of trifluoroacetic acid at approximately 50° C.
reaction suitability
reagent type: derivatization reagent
reaction type: Acylations
reagent type: derivatization reagent
reaction type: Esterifications
Safety Profile
A severe skin and eye irritant.Explosive reaction with dimethyl sulfoxide; nitric acid +1,3,5-triazine (at 36°C); nitric acid + 1,3,5-triacetylhexahydro-1,3,5-triazine (at 30°C). Incompatiblewith lithium tetrahydroaluminate. When heated todecomposit
Purification Methods
Purification by distilling over KMnO4, as for the acid above, is EXTREMELY DANGEROUS due to the possiblility of EXPLOSION. It is best purified by distilling from P2O5 slowly, and collecting the fraction boiling at 39.5o. Store it in a dry atmosphere. Highly TOXIC vapour and attacks skin, work in an efficient fume hood. [Beilstein 2 IV 469.]