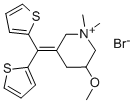
Timepidium bromide
Bezeichnung:Timepidium bromide
CAS-Nr35035-05-3
Englisch Name:Timepidium bromide
CBNumberCB9308839
SummenformelC17H22BrNOS2
Molgewicht400.4
MOL-Datei35035-05-3.mol
Timepidium bromide physikalisch-chemischer Eigenschaften
| Schmelzpunkt | 198-200° |
| storage temp. | Store at -20°C |
| Löslichkeit | Soluble in DMSO |
Gefahreninformationscode (GHS)
-
Bildanzeige (GHS)

-
Alarmwort
Warnung
-
Gefahrenhinweise
H302:Gesundheitsschädlich bei Verschlucken.
H315:Verursacht Hautreizungen.
H319:Verursacht schwere Augenreizung.
H335:Kann die Atemwege reizen.
-
Sicherheit
P261:Einatmen von Staub vermeiden.
P305+P351+P338:BEI KONTAKT MIT DEN AUGEN: Einige Minuten lang behutsam mit Wasser spülen. Eventuell vorhandene Kontaktlinsen nach Möglichkeit entfernen. Weiter spülen.
Timepidium bromide Chemische Eigenschaften,Einsatz,Produktion Methoden
-
Originator
Sesden,TANABE SEIYAKU,Japan,1976 -
Definition
ChEBI: Timepidium bromide is an organic molecular entity. -
Manufacturing Process
120 g of 5-hydroxynicotinic acid are dissolved in 1 liter of methanol. After saturating with dry-hydrogen chloride gas at 0°C, the solution is refluxed for 2 hours. Then, the solution is concentrated to dryness. The residue thus obtained is dissolved in water. The solution is neutralized with sodium bicarbonate. The precipitated crystals are collected by filtration, washed with water and then dried. 126 g of methyl 5-hydroxynicotinate are obtained. Yield: 93%. Melting point 184°C to 186°C.
460 g of methyl 5-hydroxynicotinate and 621 g of potassium carbonate are suspended in 200 ml of tetrahydrofuran-methanol (4:1). 1,134 g of dimethyl sulfate are added dropwise to the suspension in nitrogen atmosphere at room temperature. The mixture is stirred overnight at the same temperature and then filtered. The filtrate is concentrated to dryness. The residue thus obtained is mixed with 1.6 liters of methanol and 280 ml of Raney-nickel, and hydrogenated overnight in an autoclave at room temperature and at a pressure of 85 atmospheres. 200 g of Raney-nickel are added to the reaction mixture. The mixture is adjusted to pH 9.5 with triethylamine, and is further subjected to hydrogenation for 20 hours in an autoclave at 70°C and at a pressure of 100 atmospheres. Potassium carbonate and a small amount of ice are added to the reaction mixture to bring the pH to 11. The mixture is extracted with ether. After drying, the ether layer is filtered. The filtrate is evaporated to remove ether. The residue thus obtained is distilled under reduced pressure. 450 g of methyl N-methyl-5-methoxynipecotinate are obtained. Yield: 80%. Boiling point 80°C to 81°C/0.5 mm Hg. A solution of 18 g of 2-thienyl bromide in 30 ml of tetrahydrofuran is gradually added to a mixture of 2.6 g of magnesium and 80 ml of tetrahydrofuran under stirring at 50°C. The mixture is stirred for 5 hours at room temperature until the magnesium is entirely dissolved in the solution. 6.2 g of methyl N-methyl-5-methoxy-nipecotinate are added to the mixture. Then, the mixture is refluxed for 4 hours. After the reaction is completed, tetrahydrofuran is distilled off under reduced pressure. An aqueous ammonium chloride solution is added to the residue, and the solution is extracted with chloroform. The extract is dried and then evaporated to remove chloroform. The viscous oil thus obtained is recrystallized from a mixture of benzene and ether. 7 g of di-(2-thienyl)-(N-methyl-5-methoxy-3-piperidyl)- carbinol are obtained as crystals. Melting point 142°C to 146°C. 7 g of the product are dissolved in 150 ml of 10% hydrochloric acid, and the solution is heated at 80°C for 30 minutes. After the reaction is completed, the solution is basified with sodium hydroxide and then extracted with ether. The extract is washed with water, dried and evaporated to remove ether. 5 g of di- (2-thienyl)-(N-methyl-5-methoxy-3-piperidylidene)-methane are obtained as pale yellow oil.
365 mg of di-(2-thienyl)-(N-methyl-5-methoxy-3-piperidylidene)-methane are dissolved in 15 ml of ether. 1 ml of methyl bromide is added to the solution. Then, the solution is stirred overnight. The precipitated crystals are collected by filtration and recrystallized from a mixture of acetone and ether. 390 mg of di-(2-thienyl)-(N-methyl-5-methoxy-3-piperidylidene)-methane methyl bromide are obtained as colorless crystals. Melting point 198°C to 200°C. -
Therapeutic Function
Anticholinergic -
in vivo
Effects of Timepidium bromide (TB), acetylcholine (ACh) and neostigmine (Neost) on gastric and duodenal blood flow distribution are studied by the use of 131 I-labeled macroaggregated human serum albumin (MAA) in rabbits. In normal rabbits, gastric blood flow is found to be uneven in various regions of the stomach: anterior corpus (50% of total gastric blood flow) greater than posterior corpus (40%) greater than pyloric antrum (7%). Intravenous administration of Timepidium bromide (200 μg/kg) to normal rabbits produces a slight increase in total gastric blood flow, but the increase in the mucosal layer of the pyloric antrum is considerable.
Timepidium bromide Anbieter Lieferant Produzent Hersteller Vertrieb Händler.
Global(26)Suppliers
-
CONIER CHEM AND PHARMA LIMITED
Telefon +8618523575427
E-Mail sales@conier.com
-
Telefon +1-781-999-5354<br/>+1-00000000000
E-Mail marketing@targetmol.com
-
Telefon 021-58955995
E-Mail sales@medchemexpress.cn
-
Fan De(Beijing) Biotechnology Co., Ltd.
Telefon 15911056312
E-Mail liming@bio-fount.com
-
Jinan Ruisi Pharmaceutical Technology Co., LTD
Telefon 0531-69900633<br/>18340090523
E-Mail changyuan20@163.com
-
Telefon 13348960310<br/>13348960310
E-Mail 3003867561@qq.com
-
Beijing Jin Ming Biotechnology Co., Ltd.
Telefon 010-60605840<br/>15801484223;
E-Mail psaitong@jm-bio.com
-
Telefon 4008200310
E-Mail marketing@tsbiochem.com
1of2
35035-05-3, Timepidium bromide Verwandte Suche:
- Methoxydiethylborane
- Pinaveriumbromid
- 4,4'-[(2β,3α,5α,16β,17β)-3,17-Bis(acetoxy)androstan-2,16-diyl]bis[1,1-dimethylpiperazinium]dibromid
- 3,8-Diamino-1-ethyl-6-phenylphe-nantridinium-bromid
- 3-Methoxybenzylalkohol
1of4
