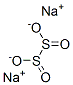
Natriumdithionit
Bezeichnung:Natriumdithionit
CAS-Nr7775-14-6
Englisch Name:Sodium dithionite
CBNumberCB1155576
SummenformelNa2O4S2
Molgewicht174.11
MOL-Datei7775-14-6.mol
Synonyma
Natriumdithionit
Natriumhypodisulfit
Dinatriumhydrosulfit
Natriumhydrosulfit
Natriumhyposulfit
Natriumdithionit physikalisch-chemischer Eigenschaften
| Schmelzpunkt | 300 °C |
| Siedepunkt | 1390°C |
| Dichte | 2.13 |
| Flammpunkt | >100°C |
| storage temp. | Store at +5°C to +30°C. |
| Löslichkeit | 250 g/L (20°C) |
| Aggregatzustand | Powder/Solid |
| Farbe | White |
| Geruch (Odor) | None or slight scent of sulfur dioxide |
| PH | 5.5-8.5 (50g/l, H2O, 20℃) |
| Wasserlöslichkeit | 250 g/L (20 ºC) |
| Sensitive | Moisture Sensitive |
| Merck | 14,8626 |
| Stabilität | Stable, but air sensitive. Incompatible with strong acids, strong oxidizing agents, water, moisture. |
| LogP | -2.756 (est) |
| CAS Datenbank | 7775-14-6(CAS DataBase Reference) |
| EPA chemische Informationen | Sodium hydrosulfite (7775-14-6) |
| Kennzeichnung gefährlicher | Xn |
| R-Sätze: | 7-22-31 |
| S-Sätze: | 26-28-43-7/8-43E-28A |
| RIDADR | UN 1384 4.2/PG 2 |
| WGK Germany | 1 |
| F | 1-10 |
| Selbstentzündungstemperatur | >200 °C |
| TSCA | Yes |
| HazardClass | 4.2 |
| PackingGroup | II |
| HS Code | 28311010 |
| Giftige Stoffe Daten | 7775-14-6(Hazardous Substances Data) |
| Toxizität | LD50 orally in Rabbit: 2500 mg/kg |
Gefahreninformationscode (GHS)
-
Bildanzeige (GHS)


-
Alarmwort
Achtung
-
Gefahrenhinweise
H251:Selbsterhitzungsfähig; kann in Brand geraten.
H302:Gesundheitsschädlich bei Verschlucken.
H319:Verursacht schwere Augenreizung.
-
Sicherheit
P235:Kühl halten
P264:Nach Gebrauch gründlich waschen.
P264:Nach Gebrauch gründlich waschen.
P270:Bei Gebrauch nicht essen, trinken oder rauchen.
P280:Schutzhandschuhe/Schutzkleidung/Augenschutz tragen.
P301+P312:BEI VERSCHLUCKEN: Bei Unwohlsein GIFTINFORMATIONSZENTRUM/Arzt/... (geeignete Stelle für medizinische Notfallversorgung vom Hersteller/Lieferanten anzugeben) anrufen.
P305+P351+P338:BEI KONTAKT MIT DEN AUGEN: Einige Minuten lang behutsam mit Wasser spülen. Eventuell vorhandene Kontaktlinsen nach Möglichkeit entfernen. Weiter spülen.
Sodium dithionite Chemische Eigenschaften,Einsatz,Produktion Methoden
-
ERSCHEINUNGSBILD
WEISSES KRISTALLINES PULVER. -
CHEMISCHE GEFAHREN
Die Substanz zersetzt sich beim Erhitzen oberhalb 100°C unter Bildung von giftigen Rauchen mit Schwefeloxiden. Starkes Reduktionsmittel. Reagiert mit Oxidationsmitteln. Die Substanz zersetzt sich bei Kontakt mit Säuren unter Bildung von giftigen Gasen. Kontakt mit Wasser, Feuchtigkeit oder feuchter Luft kann spontane Entzündung verursachen. -
ARBEITSPLATZGRENZWERTE
MAK nicht festgelegt.
TLV nicht festgelegt.
-
AUFNAHMEWEGE
Aufnahme in den Körper durch Verschlucken. -
INHALATIONSGEFAHREN
Eine gesundheitsschädliche Partikelkonzentration in der Luft kann schnell erreicht werden beim Dispergieren, vor allem als Pulver. -
WIRKUNGEN BEI KURZZEITEXPOSITION
WIRKUNGEN BEI KURZZEITEXPOSITION:
Die Substanz reizt die Augen und die Atemwege. -
LECKAGE
Persönliche Schutzausrüstung: Atemschutzgerät mit Partikelfilter und Filter für saure Gase entsprechend der Arbeitsplatzkonzentration des Stoffes. Verschüttetes Material in abgedeckten Behältern sammeln. Reste sorgfältig sammeln. An sicheren Ort bringen. NICHT mit Sägemehl oder anderen brennbaren Absorptionsmitteln binden. NICHT in die Umwelt gelangen lassen. -
R-Sätze Betriebsanweisung:
R7:Kann Brand verursachen.
R22:Gesundheitsschädlich beim Verschlucken.
R31:Entwickelt bei Berührung mit Säure giftige Gase. -
S-Sätze Betriebsanweisung:
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S28:Bei Berührung mit der Haut sofort abwaschen mit viel . . . (vom Hersteller anzugeben).
S43:Zum Löschen . . . (vom Hersteller anzugeben) verwenden (wenn Wasser die Gefahr erhöht, anfügen: "Kein Wasser verwenden").
S7/8:Behälter trocken und dicht geschlossen halten. -
Chemische Eigenschaften
White solid -
Verwenden
Sodium hydrosulfite is used as a reducing agent in aqueous solutions, sulfonating agent , chelating agent and decolorizing agent in organic reactions. It finds application in water treatment, gas purification, cleaning, leather, polymers, photography, and many others. It is involved in chemical enhanced oil recovery to stabilize polyacrylamide polymers against radical degradation in the presence of iron. It plays an important role to determine the iron content in soil chemistry. -
Verwenden
Sodium dithionite is a reducing agent for vat dyes,reducing hair bleaching agents,vat dyes printing auxiliaries, silk scouring and bleaching agents, coloring agents and objects stripping vat cleaning agent, particularly in dyeing with indigo and vat dyes; bleaching soaps, straw; removing dyes from dyed fabrics. -
Definition
ChEBI: An inorganic sodium salt that is the disodium salt of dithionous acid. -
Vorbereitung Methode
An alternative route for dithionite production is the reduction of sodium bisulfite with sodium borohydride. Sodium borohydride is obtained by reacting boron trimethyl ester, B(OCH3)3, with sodium hydride, NaH. The resulting product is hydrolyzed with water, and methanol is evaporated. An alkaline, aqueous solution is obtained, containing about 12% NaBH4 and 40% NaOH. This solution is commercially available. Reaction to dithionite is made on-site by adding sulfur dioxide and some additional caustic soda. Storages, handling and mixing of sulfur dioxide and of the Borol® liquid are not everywhere cost competitive.
NaBH4+8NaOH+8SO2 → 4Na2S2O4+NaBO2+6H2O. -
synthetische
Sodium dithionite is today produced mainly from sodium formate and sulfur dioxide. Zinc dithionite, prepared on-site from sulfur dioxide and zinc dust, was formerly more important than the sodium salt. The heavy metal zinc ended up in the effluent, which caused environmental problems. Today in pulp mills on-site production of zinc dithionite is no longer practiced. The number of plants using sodium amalgam and sulfur dioxide for dithionite production is decreasing with the number of amalgam cells in electrolysis. On-site generation of dithionite from alkaline sodium borohydride solution, sodium bisulfite, and sulfur dioxide is used by some mills.
Sodium dithionite is available as crystalline powder (&90% Na2S2O4) or as a refrigerated 150 g/L solution stabilized with alkali. The white crystals can decompose on heating. Low alkalinity, pH8 to 13, stabilizes dithionite solution at low temperature.
These solutions should be kept well below 10 °C with the exclusion of air. In presence of air, oxidation rapidly yields sulfate and sulfite.
The powder product prepared via the amalgam route dissolves into an alkaline solution as it contains sodium carbonate and sulfite. This can trigger the precipitation of calcium carbonate (water hardness) in the solution tank. Therefore, it is recommended to add small amounts of chelants during the dissolution step. (Chelants are not required to stabilize the bleaching reaction.) Sodium dithionite prepared by the formate route dissolves into a slightly acidic solution. Dithionite powder can ignite if exposed to high humidity or elevated temperature. -
Allgemeine Beschreibung
Sodium dithionite is a whitish to light yellow crystalline solid having a sulfur dioxide-like odor. Sodium dithionite spontaneously heats on contact with air and moisture. This heat may be sufficient to ignite surrounding combustible materials. Sodium dithionite is soluble in water. Under prolonged exposure to fire or intense heat containers of Sodium dithionite may violently rupture. Sodium dithionite is used in dyeing and to bleach paper pulp. -
Reaktivität anzeigen
Inorganic reducing agents, such as Sodium dithionite, react with oxidizing agents to generate heat and products that may be flammable, combustible, or otherwise reactive. Their reactions with oxidizing agents may be violent. Sulfites and hydrosulfites (dithionites) can react explosively with strong oxidizing agents (sodium chlorite). Sulfites generate gaseous sulfur dioxide in contact with oxidizing acids and nonoxidizing acids. -
Hazard
Fire risk in contact with moisture. To extin- guish fires, flood the reacting mass with water. -
Health Hazard
Fire will produce irritating, corrosive and/or toxic gases. Inhalation of decomposition products may cause severe injury or death. Contact with substance may cause severe burns to skin and eyes. Runoff from fire control may cause pollution. -
Brandgefahr
Flammable/combustible material. May ignite on contact with moist air or moisture. May burn rapidly with flare-burning effect. Some react vigorously or explosively on contact with water. Some may decompose explosively when heated or involved in a fire. May re-ignite after fire is extinguished. Runoff may create fire or explosion hazard. Containers may explode when heated. -
Flammability and Explosibility
Not classified -
Sicherheitsprofil
Toxic and an irritant. An allergen. Flammable when exposed to heat or flame. Ignites on contact with water or sodium chlorite. To extinpsh fires, flood the reacting mass with water. Decomposes violently when heated to 19OOC and emits toxic fumes of SOx and NazO -
Lager
Sodium dithionite crystals are available in steel containers (1 or 2 tons) or in steel drums (200 kg). Because of the danger of spontaneous ignition in humid air, sodium dithionite must be stored under dry and cool conditions. The sites for the preparation of dithionite solutions must permit handling without risks, high humidity should be avoided and remote fire control should be available.
Commercial dithionite products are classified as self-igniting hazardous goods (Class 4.2, UN 1384). Local rules for transportation and storage must be obeyed. -
Properties and Applications
INDEX/GRADE
SH-90
SH-88
SH-85
CONTENT
90%
88%
85%
Na 2 S 2 O 4
≥90%
≥88%
≥85%
Fe
≤20ppm
≤20ppm
≤20ppm
Zinc (Zn)
≤1ppm
≤1ppm
≤1ppm
Other heavy metal
(calculated as Pb)
≤1ppm
≤1ppm
≤1ppm
Water Insolubles
≤0.05%
≤0.05%
≤0.05%
Shelf Life(month)
12 <
Sodium dithionite Upstream-Materialien And Downstream Produkte
Upstream-Materialien
1of3
Downstream Produkte
- Natriumdithionat
- Vitamin B2
- 3,7-Dihydro-8-methyl-1H-purin-2,6-dion
- 4-Aminobenzylalkohol
- 4-AMINORESORCINOL HYDROCHLORIDE
- 4,4'-Diaminodiphenylaminsulfat
- 5-Fluor-1H-indol-2-carbonsure
- Natriumsalicylat
- 5-Amino-1,2,3,4-tetrahydrophtha-lazin-1,4-dion
- (2,4-DIAMINOPTERIDIN-6-YL)METHANOL HYDROCHLORIDE HYDRATE
- 2,3,5-Trimethylhydrochinon
- Pyrimidin-4,5,6-triyltriamin
- 4-amino-1-naphthol
- 3-Hydroxy-2'-methoxy-2-naphthanilid
- 3,7-Bis(diethylamino)phenoxazin-5-iumchlorid
- 4-(1,2,3-THIADIAZOL-4-YL)ANILINE
- 4-Azaindole
- Bis(5,6-diaminouracil)sulfat
- TRIFLUOROMETHYL SULFINYL CHLORIDE
- 7-Amino-1H-triazolo[4,5-d]pyrimidin
- Pyrimidin-4,5,6-triaminsulfat (1:1)
- 2,2'-Ethandiylidendinitrilodiphenol
- Riboflavin
- 8-BROMO-3-METHYL-3,7-DIHYDRO-PURINE-2,6-DIONE
- 5-Hydroxyanthranilic acid
- 5-Amino-1H-triazolo[4,5-d]pyrimidin-7-ol
- 2-Hydroxy-2',5'-dimethoxydibenzofuran-3-carboxanilid
- 3,7-Dihydro-3-methyl-1H-purin-2,6-dion
- 3-(3-AMINOPHENYL)-2H-CHROMEN-2-ONE
- (2-CYANO-3-PYRIDINYL)CARBAMIC ACID, 1,1-DIMETHYL ETHYL ESTER
1of8
Natriumdithionit Anbieter Lieferant Produzent Hersteller Vertrieb Händler.
Global(604)Suppliers
India
Region
Argentina 3 Australia 3 Belgium 6 Brazil 2 Canada 6 China 286 Costa Rica 1 Czech Republic 1 Denmark 1 Egypt 2 Europe 3 France 3 Germany 16 Hungary 1 India 103 Italy 9 Japan 10 Malaysia 2 Mexico 8 Russia 2 Singapore 3 Slovakia 1 South Korea 3 Spain 2 Switzerland 3 Thailand 2 The Netherlands 5 United Arab Emirates 1 United Kingdom 21 United States 95-
Telefon --
E-Mail rajeevm@siloxindia.com
-
Telefon --
E-Mail demosha@gmail.com
-
Telefon --
E-Mail bd@adichemsol.com
-
Telefon --
E-Mail st@rubexco.com
-
Telefon --
E-Mail chem@tcpindia.com
-
Telefon --
E-Mail rekha.bijoykumar@merckgroup.com
-
Telefon --
E-Mail delent@vsnl.com
-
Panoli Intermediates (India) Pvt., Limited. (Kutch Chemical Industries Ltd.)
Telefon --
E-Mail jkpatel@kcil.co.in
-
Telefon --
E-Mail jsk.chemicals@gmail.com
-
TCI Chemicals (India) Pvt. Ltd.
Telefon --
E-Mail sales-in@tcichemicals.com
-
Telefon --
E-Mail info@chemit.in
-
Telefon --
E-Mail sales@trivenichemical.com
-
Telefon --
E-Mail tech@alfa.com
-
Telefon --
E-Mail info@ottokemi.com
-
Telefon --
E-Mail supremeind@mtnl.net.in
-
Piyush Pharmachem (I) Pvt. Ltd.
Telefon --
E-Mail info@piyushpharmachem.com
-
Telefon --
E-Mail shrisang@vsnl.com
-
Telefon --
E-Mail demosha@gmail.com
-
Telefon --
E-Mail
-
Telefon --
E-Mail info@aadishimpex.com
-
Telefon --
E-Mail info@pure-chemical.com
-
Telefon --
E-Mail kunal@aimchemicals.in
-
Telefon --
E-Mail
-
Transpek-Silox Industry Pvt. Ltd.
Telefon --
E-Mail rajeevm@tsilbrd.com
-
Telefon --
E-Mail
-
Telefon --
E-Mail kareemali@aljamiaenterprises.com
-
Telefon --
E-Mail
-
Panoli Intermediates (India) Pvt., Limited. (Kutch Chemical Industries Ltd.)
Telefon --
E-Mail snpatel@kcil.co.in
-
Telefon --
E-Mail
-
Telefon --
E-Mail
-
Telefon --
E-Mail bd@adichemsol.com
-
Telefon --
E-Mail behl100@rediffmail.com
-
Telefon --
E-Mail info@desmoexports.com
-
Telefon --
E-Mail @lobachemie.com
-
Telefon --
E-Mail cheminquiry@gmail.com
-
Telefon --
E-Mail sales@adventchembio.com
-
New Bright Alloychem Pvt., Ltd.
Telefon --
E-Mail nbacpl@gmail.com
-
Dwarka Dass Chemical Co Pvt. Ltd.
Telefon --
E-Mail info@dwarkadasschemical.com
-
Telefon --
E-Mail
-
Artichempharma (Aarti Dye Chem)
Telefon --
E-Mail info@aartichempharma.com
-
Telefon --
E-Mail
-
Telefon --
E-Mail delent@vsnl.com
-
Telefon --
E-Mail info@amayaint.com
-
Telefon --
E-Mail
-
Telefon --
E-Mail
-
Praful Venture Pvt., Ltd. (Praful Group of Companies)
Telefon --
E-Mail harish.praful@gmail.com
-
Telefon --
E-Mail shantichemicals@gmail.com
-
Telefon --
E-Mail info@veeraj.in
-
Chemrich Fine Chemicals Pvt., Ltd.
Telefon --
E-Mail info@chemrichfine.com
-
Telefon --
E-Mail rupesh@bhimanigroup.com
1of10
7775-14-6, Sodium dithionite Verwandte Suche:
