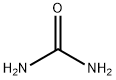
Мочевина
- английское имяUrea
- CAS №57-13-6
- CBNumberCB5853861
- ФормулаCH4N2O
- мольный вес60.06
- EINECS200-315-5
- номер MDLMFCD00008022
- файл Mol57-13-6.mol
| Температура плавления | 132-135 °C(lit.) |
| Температура кипения | 332.48°C (estimate) |
| плотность | 1.335 g/mL at 25 °C(lit.) |
| Плотность накопления | 720-760kg/m3 |
| давление пара | <0.1 hPa (20 °C) |
| показатель преломления | n |
| температура хранения | 2-8°C |
| растворимость | H2O: 8 M at 20 °C |
| форма | powder |
| пка | 0.10(at 25℃) |
| цвет | white |
| Удельный вес | 1.335 |
| Запах | almost odorless |
| РН | 8.0-10.0 (20℃, 8M in H2O) |
| Биологические источники | synthetic |
| Растворимость в воде | 1080 g/L (20 ºC) |
| λмакс | λ: 260 nm Amax: 0.03 λ: 280 nm Amax: 0.02 |
| Мерк | 14,9867 |
| БРН | 635724 |
| Диэлектрическая постоянная | 3.5(Ambient) |
| Стабильность | Substances to be avoided include strong oxidizing agents. Protect from moisture. |
| ИнЧИКей | XSQUKJJJFZCRTK-UHFFFAOYSA-N |
| LogP | -1.660 (est) |
| FDA 21 CFR | 184.1923; 175.300; 177.1200; 310.545; 573.220; 573.940 |
| Вещества, добавляемые в пищу (ранее EAFUS) | UREA |
| Специальный комитет по веществам GRAS | Urea |
| Справочник по базе данных CAS | 57-13-6(CAS DataBase Reference) |
| Рейтинг продуктов питания EWG | 1-3 |
| Словарь онкологических терминов NCI | carbamide; urea |
| FDA UNII | 8W8T17847W |
| Код УВД | B05BC02,D02AE01,D02AE51 |
| Справочник по химии NIST | Urea(57-13-6) |
| Система регистрации веществ EPA | Urea (57-13-6) |
| Поглощение | ≤0.06 at 260nm at 5M ≤0.06 at 280nm at 5M |
| UNSPSC Code | 41171614 |
| NACRES | NB.22 |
| Коды опасности | Xn,Xi | |||||||||
| Заявления о рисках | 36/37/38-40-38 | |||||||||
| Заявления о безопасности | 26-36-24/25-37 | |||||||||
| РИДАДР | Not regulated | |||||||||
| WGK Германия | 1 | |||||||||
| RTECS | YR6250000 | |||||||||
| TSCA | Yes | |||||||||
| кода HS | 31021010 | |||||||||
| Банк данных об опасных веществах | 57-13-6(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 orally in Rabbit: 8471 mg/kg LD50 dermal Rat 8200 mg/kg | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)

-
сигнальный язык
предупреждение
-
оператор предупредительных мер
P264:После работы тщательно вымыть кожу.
P305+P351+P338:ПРИ ПОПАДАНИИ В ГЛАЗА: Осторожно промыть глаза водой в течение нескольких минут. Снять контактные линзы, если Вы ими пользуетесь и если это легко сделать. Продолжить промывание глаз.
P337+P313:Если раздражение глаз не проходит обратиться за медицинской помощью.
Мочевина химические свойства, назначение, производство
Описание
Urea is a stable highly water-soluble compound of high nitrogen content (47%), with good storage properties that make it the most commonly used nitrogen fertilizer. The synthesis process has remained essentially unchanged since it was first developed by the BASF Corporation in 1922. In this process, liquid ammonia is reacted with carbon dioxide to produce ammonium carbamate, which is then dehydrated to form urea. The reactions are:2NH3 + CO2 ===? NH2·CO2·NH4
NH2·CO2·NH4 ===? (NH2)2CO + H2O
Химические свойства
Urea,CO(HN2)2, also known as carbamide, is a white crystalline powder that has a melting point of l32.7 °C (270 °F). It is a natural product of animal protein metabolism and is the chief nitrogen constituent of urine. Commercially, urea is produced by the reaction of ammonia and carbon dioxide. It is soluble in water, alcohol, and benzene.Вхождение
The compound was discovered by Hilaire Rouelle in 1773 as a constituent of urine.История
Urea has the distinction of being the first synthesized organic compound. Until the mid-18th century, scientists believed organic compounds came only from live plants and animals. The first serious blow to the theory of vitalism, which marked the beginning of modern organic chemistry, occurred when Friedrich W?hler (1800 1882) synthesized urea from the two inorganic substances, lead cyanate and ammonium hydroxide: Pb(OCN)2 + 2NH4OH→2(NH2)2CO + Pb(OH)2. W?hler's discoveries on urea occurred while he was studying cyanates; he was attempting to synthesize ammonium cyanate when he discovered crystals of urea in his samples. He first prepared urea in 1824, but he did not identify this product and report his findings until 1828. W?hler's synthesis of urea signaled the birth of organic chemistry.Использование
Urea is a physiological regulator of nitrogen excretion in mammals; synthesized in the liver as an end-product of protein catabolism and excreted in urine. Also occurs normally in skin. Emollient; diu retic.Методы производства
Urea is an important industrial compound. The synthesis of urea was discovered in 1870.Commercial production of urea involves the reaction of carbon dioxide and ammonia at highpressure and temperature to produce ammonium carbamate. Ammonium carbamate is thendehydrated to produce urea (Figure 96.1). The reaction uses a molar ratio of ammonia tocarbon dioxide that is approximately 3:1 and is carried out at pressures of approximately 150atmospheres and temperatures of approximately 180°C.Определение
A white crystalline compound made from ammonia and carbon dioxide. It is used in the manufacture of urea–formaldehyde (methanal) resins. Urea is the end product of metabolism in many animals and is present in urine.Показания
Urea-containing preparations have a softening and moisturizing effect on the stratum corneum and, at times, may provide good therapy for dry skin and the pruritus associated with it. They appear to have an antipruritic effect apart from their hydrating qualities. Urea compounds disrupt the normal hydrogen bonds of epidermal proteins; therefore, their effect in dry hyperkeratotic diseases such as ichthyosis vulgaris and psoriasis is not only to make the skin more pliable but also to help remove adherent scales. Lactic acid also has a softening and moisturizing effect on the stratum corneum.Urea 40% ointment may be useful in removing hypertrophic or dystrophic psoriatic nails. Subsequent topical therapy to the denuded nail bed and proximal nail fold may result in regrowth of ‘‘normal’’ nails in half of those treated.
Биологические функции
The use of urea (Ureaphil, Urevert) has declined in recent years owing both to its disagreeable taste and to the increasing use of mannitol for the same purposes. When used to reduce cerebrospinal fluid pressure, urea is generally given by intravenous drip. Because of its potential to expand the extracellular fluid volume, urea is contraindicated in patients with severe impairment of renal, hepatic, or cardiac function or active intracranial bleeding.Общее описание
Solid odorless white crystals or pellets. Density 1.335 g /cc. Noncombustible.Реакции воздуха и воды
Water soluble.Профиль реактивности
Urea is a weak base. Reacts with hypochlorites to form nitrogen trichloride which explodes spontaneously in air [J. Am. Chem. Soc. 63:3530-32]. Same is true for phosphorus pentachloride. Urea reacts with azo and diazo compounds to generate toxic gases. Reacts with strong reducing agents to form flammable gases (hydrogen). The heating of improper stoichiometric amounts of Urea and sodium nitrite lead to an explosion. Heated mixtures of oxalic acid and Urea yielded rapid evolution of gases, carbon dioxide, carbon monoxide and ammonia (if hot, can be explosive). Titanium tetrachloride and Urea slowly formed a complex during 6 weeks at 80°C., decomposed violently at 90°C., [Chem. Abs., 1966, 64, 9219b]. Urea ignites spontaneously on stirring with nitrosyl perchlorate, (due to the formation of the diazonium perchlorate). Oxalic acid and Urea react at high temperatures to form toxic and flammable ammonia and carbon monoxide gasses, and inert CO2 gas [Von Bentzinger, R. et al., Praxis Naturwiss. Chem., 1987, 36(8), 41-42].Угроза здоровью
May irritate eyes.Пожароопасность
Behavior in Fire: Melts and decomposes, generating ammonia.Сельскохозяйственное использование
Fertilizer, Fungicide: Used in fertilizers and animal feeds, as a fungicide, in the manufacture of resins and plastics, as a stabilizer in explosives and in medicines, and others. Urea is used to protect against frost and is used in some pesticides as an inert ingredient as a stabilizer, as an inhibitor and as an intensifier for herbicides. Registered for use in EU countries . Registered for use in the U.S.Торговое название
PRESPERSION, 75 UREA®; SUPERCEL 3000®; UREAPHIL®; UREOPHIL®; UREVERT®; VARIOFORM II®Профиль безопасности
Moderately toxic by intravenous and subcutaneous routes. Human reproductive effects by intraplacental route: ferthty effects. Experimental reproductive effects. Human mutation data reported. A human skin irritant. Questionable carcinogen with experimental carcinogenic and neoplastigenic data. Reacts with sodium hypochlorite or calcium hypochlorite to form the explosive nitrogen trichloride. Incompatible with NaNO2, P2Cl5, nitrosyl perchlorate. Preparation of the 15N-labeled urea is hazardous. When heated to decomposition it emits toxic fumes of NOx.Возможный контакт
Urea is used in ceramics, cosmetics, paper processing; resins, adhesives, in animal feeds; in the manufacture of isocyanurates; resins, and plastics; as a stabilizer in explosives; in medicines; anticholelithogenic, and others.Экологическая судьба
Terrestrial FateUrea is expected to have very high mobility in soil. Urea is not expected to volatilize from dry soil surfaces based on its vapor pressure. Various field and laboratory studies have demonstrated that urea degrades rapidly in most soils. Urea is rapidly hydrolyzed to ammonium ions through soil urease activity, which produces volatile gases, that is, ammonia and carbon dioxide. However, the rate of hydrolysis can be much slower, depending on the soil type, moisture content, and urea formulation.
Aquatic Fate
Urea is not expected to adsorb to suspended solids and sediments. Volatilization from water surfaces is not expected. Urea is rapidly hydrolyzed to ammonia and carbon dioxide in environmental systems by the extracellular enzyme urease, which originates from microorganisms and plant roots.
Atmospheric Fate
According to a model of gas/particle partitioning of semivolatile organic compounds in the atmosphere, urea, which has a vapor pressure of 1.2×10-5mm Hg at 251°C, will exist in both the vapor and particulate phases in the ambient atmosphere. Vapor-phase urea is degraded in the atmosphere by reaction with photochemically produced hydroxyl radicals; the half-life for this reaction in air is estimated to be 9.6 days.
Метаболизм
The high analysis and good handling properties of urea have made it the leading nitrogen fertilizer, both as a source of nitrogen alone or when compounded with other materials in mixed fertilizers. Although an excellent source of nitrogen, urea can present problems unless properly managed; due to its rapid hydrolysis to ammonia, significant volatilization loss of this may occur if prilled or granular urea is applied to and left on the soil surface without timely incorporation. Mixtures of urea and ammonium nitrate for use in mixed fertilizers are also more highly hygroscopic than ammonium nitrate itself.Методы очистки
Crystallise urea twice from conductivity water using centrifugal drainage and keeping the temperature below 60o. The crystals are dried under vacuum at 55o for 6hours. Levy and Margouls [J Am Chem Soc 84 1345 1962] prepared a 9M solution in conductivity water (keeping the temperature below 25o) and, after filtering through a medium-porosity glass sinter, added an equal volume of absolute EtOH. The mixture was set aside at -27o for 2-3 days and filtered cold. The precipitate was washed with a small amount of EtOH and dried in air. Crystallisation from 70% EtOH between 40o and -9o has also been used. Ionic impurities such as ammonium isocyanate have been removed by treating the concentrated aqueous solution at 50o with Amberlite MB-1 cation-and anion-exchange resin, and allowing it to crystallise on evaporation. [Benesch et al. J Biol Chem 216 663 1955.] It can also be crystallised from MeOH or EtOH, and is dried under vacuum at room temperature. [Beilstein 3 H 42, 3 I 19, 3 II 35, 3 III 80.]Несовместимости
Violent reaction with strong oxidizers, chlorine, permanganates, dichromates, nitrites, inorganic chlorides; chlorites, and perchlorates. Contact with hypochlorites can result in the formation of explosive compounds.Утилизация отходов
Controlled incineration in equipment containing a scrubber or thermal unit to reduce nitrogen oxide emissions.Мочевина запасные части и сырье
сырьё
1of3
запасной предмет
- Reactive Black KN-BN
- ureaformaldelyde resin UF
- 5-карбэтоксиурацил
- 2,4-DICHLOROTHIENO[3,2-D]PYRIMIDINE
- DIHYDROTHYMINE
- Nitrofurazone
- Tanning agent for white leather
- synthetic carbamider ring tanning agent No.1
- 1-(2-аминоэтил)имидазолидин-2-он
- DIRECT FAST BLACK G
- 2,4-Dichlorothieno[3,2-d]pyrimidine
- 2,6-Дихлор-4,8-дипиперидинопиримидино[5,4-d]пиримидин
- biodegrddable finishing agent for fabric
- BIUREA
- 1-(1,3-DIHYDRO-1-OXOISOBENZOFURAN-3-YL)UREA
- N, N ”- (изобутилиден) димочевина
- 1-Methyl-1-nitrosourea
- 4,5-дигидрокси-1,3-бис(гидроксиметил)имидазолидин-2-он
- 3-Hydroxy-1-phenyl-1,2,4-triazole
- ДИХЛОРИЗОЦИАНУРОВАЯ КИСЛОТА
- ЦИМОКСАНИЛ
- 2,4,6,8-Тетрагидрокси-пиримидо-(5,4D)пиримидин
- Мочевина - перекись водорода
- 5- (втор-пентил) барбитуровая кислота
- Amino moulding plastic
1of8
Мочевина поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия | |
|---|---|---|---|---|---|
| +86-18168774353 | China | 248 | 58 | ||
| +86-17331933971 +86-17331933971 |
China | 2472 | 58 | ||
| +1-+1(833)-552-7181 | United States | 57505 | 58 | ||
| +1-+1(833)-552-7181 | United States | 52924 | 58 | ||
| +86-15532196582 +86-15373005021 |
China | 3007 | 58 | ||
| +8619931165850 | China | 1000 | 58 | ||
| +86-371-86557731 +86-13613820652 |
China | 20259 | 58 | ||
| +8615713292910 | China | 341 | 58 | ||
| +86-17736087130 +86-18633844644 |
China | 994 | 58 | ||
| +8619933239880 | China | 861 | 58 |