4-Bromoaniline: Overview, Metabolism and Preparation Method
General Description
4-Bromoaniline, a versatile organic compound with aromatic and amine properties, is utilized in various applications such as dye, pigment, pharmaceutical, and agrochemical synthesis. Despite its significance, caution must be exercised due to its toxic and carcinogenic nature, necessitating proper handling and storage procedures. The metabolism of 4-bromoaniline in bile-cannulated rats was extensively studied, revealing the formation of brominated metabolites in urine and bile samples. Advanced analytical techniques like ICPMS, HPLC-ICPMS, and HPLC-oaTOFMS were instrumental in characterizing the metabolic fate of 4-bromoaniline. Furthermore, the preparation method of 4-Bromoaniline involves the amination of Bromobenzene using Hydrazine hydrate and Iron oxide Carbon as catalysts in Ethanol, resulting in a 100% yield of the desired product. This synthesis method highlights the importance of controlled reactions in organic chemistry for obtaining specific compounds efficiently.

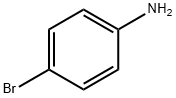
Figure 1. 4-Bromoaniline
Overview
4-Bromoaniline, also known as p-bromoaniline, is an organic compound belonging to the class of arylamines. It features a benzene ring substituted with a bromine atom at the para position and an amino group. This compound is a white to yellowish crystalline solid with a distinct odor. Chemically, it exhibits both aromatic and amine properties, making it reactive towards a variety of chemical reactions. 4-Bromoaniline finds various applications in organic synthesis, serving as a precursor for the preparation of dyes, pigments, pharmaceuticals, and agrochemicals. Its reactivity towards electrophiles allows for the introduction of diverse functionalities onto the benzene ring, leading to the synthesis of complex organic molecules. However, it should be handled with caution due to its potential toxicity and carcinogenicity. In terms of safety, proper protective measures should be taken when handling 4-Bromoaniline, including wearing protective clothing and eyewear, and working in a well-ventilated area. Additionally, it should be stored away from heat, flames, and oxidizing agents to prevent potential hazards. Overall, 4-Bromoaniline is an important intermediate in organic chemistry with a wide range of applications but requires careful handling due to its hazardous nature. 1
Metabolism
The metabolism of 4-bromoaniline in bile-cannulated rats was studied using advanced analytical techniques including ICPMS, HPLC-ICPMS, and HPLC-oaTOFMS. Following the administration of 4-bromoaniline to rats, approximately 90% of the dose was recovered in urine and bile within 48 hours. HPLC-ICPMS was used to detect and profile the major metabolites excreted in urine and bile. The analysis revealed the presence of numerous brominated metabolites in both urine and bile samples. HPLC-oaTOFMS further identified a significant number of brominated metabolites in urine and bile using negative ESI mode. Despite the detection of a large number of bromine-containing metabolites, the profiles were primarily dominated by a few major components. In urine, 4-bromoaniline O-sulfate accounted for a significant portion of the total metabolites detected, while in bile, a hydroxylated N-acetyl compound was the major metabolite. The combination of ICPMS, HPLC-ICPMS, and HPLC-oaTOFMS allowed for a comprehensive characterization of the metabolic fate of 4-bromoaniline in bile-cannulated rats without the use of radiolabeled tracers. This study provides valuable insights into the metabolism of 4-bromoaniline and demonstrates the effectiveness of these analytical techniques in studying xenobiotic metabolism. 2
Preparation Method
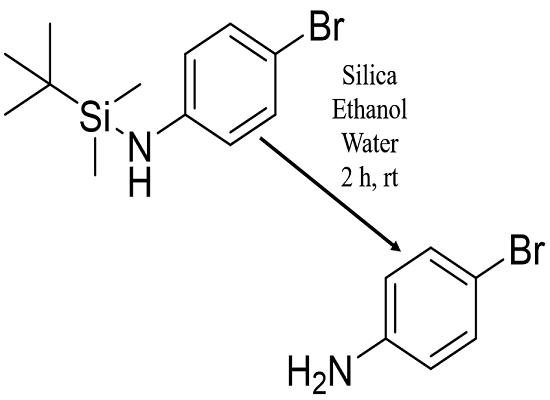
The preparation method for 4-Bromoaniline involves the conversion of Bromobenzene into 4-Bromoaniline with a yield of 100%. The reaction requires the use of Hydrazine hydrate (1:1) as the reagent and Iron oxide (Fe2O3) Carbon as the catalyst, with Ethanol as the solvent. To carry out the reaction, first, Fe-500-1h (0.025 mmol) is charged into a dried round-bottomed flask with 5 mL ethanol. Then, 102 μL of 1-bromo-4-nitrobenzene (1 mmol) and 200 μL of hydrazine hydrate (4 mmol) are added to the mixture. Next, a reflux condenser is connected to the flask, and the mixture is stirred vigorously under refluxing at 85 °C for 1.25 hours. This process results in the amination of aromatic compounds, leading to the formation of 4-Bromoaniline as the final product. 3
Reference
1. 4-Bromoaniline. National Center for Biotechnology Information. 2024; PubChem Compound Summary for CID 7807.
2. Duckett C, McCullagh M, Smith C, Wilson ID. The metabolism of 4-bromoaniline in the bile-cannulated rat: application of ICPMS ((79/81)Br), HPLC-ICPMS & HPLC-oaTOFMS. Xenobiotica. 2015; 45(8): 672-680.
3. Li Y, Zhou YX, Ma X, Jiang HL. A metal-organic framework-templated synthesis of γ-Fe2O3 nanoparticles encapsulated in porous carbon for efficient and chemoselective hydrogenation of nitro compounds. Chem Commun (Camb). 2016; 52(22): 4199-4202.
Related articles And Qustion
Lastest Price from 4-Bromoaniline manufacturers
US $1.00/kg2025-03-14
- CAS:
- 106-40-1
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $20.00-8.00/kg2025-03-07
- CAS:
- 106-40-1
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 10 tons