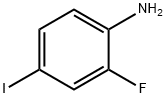
The general procedure for the synthesis of 2-fluoro-4-iodoaniline from o-fluoroaniline is as follows: o-fluoroaniline (54 g, 486 mmol) was added to an aqueous solution (250 mL) of sodium bicarbonate (41 g, 486 mmol) with vigorous stirring. The suspension was heated to 60 °C in an oil bath and iodine (123 g, 486 mmol) was added in batches. After addition, the dark-colored mixture was continued to be stirred at 60°C for 3 hours. After completion of the reaction, it was cooled to room temperature and dichloromethane (300 mL) was added, followed by saturated sodium bisulfite solution (300 mL). The two-phase system was stirred vigorously for 10 minutes. The mixture was transferred to a 2L separatory funnel and the organic layer was separated. The aqueous layer was further extracted with dichloromethane (3 x 200 mL) and the combined organic phases were washed with brine (200 mL) and dried over anhydrous sodium sulfate. After filtration, the solvent was removed under reduced pressure to give a black crystalline solid. Hexane (300 mL) was added to the solid and the mixture was heated to reflux. A hexane solution was poured from the black insoluble syrup. Upon cooling, the product crystallized from hexane as fine yellow needles. Final isolation afforded 65 g (274 mmol) of 2-fluoro-4-iodoaniline as a fine yellow solid in 56% yield with a melting point of 53°C. 1H NMR (400 MHz, CDCl3) δ 8.1 (t, J=8Hz, 1H), 7.4 (d, J=6Hz, 1H), 7.2 (d, J=6Hz, 1H).