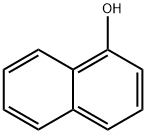
1-Naphthol, or α-naphthol, is a fluorescent organic compound with the formula C10H7OH. It is a white solid. It is an isomer of 2-naphthol differing by the location of the hydroxyl group on the naphthalene ring. The naphthols are naphthalene homologues of phenol, with the hydroxyl group being more reactive than in the phenols. Both isomers are soluble in simple alcohols, ethers, and chloroform. They are precursors to a variety of useful compounds. Naphthols (both 1 and 2 isomers) are used as biomarkers for livestock and humans exposed to polycyclic aromatic hydrocarbons.
α-naphtol, combined with epichlorhydrine and sodium hydroxide to form alpha-naphtyl glycidyl ether, caused sensitization in one of three workers in a chemical plant.
1-Naphthol is a light grey to brown solid with an unpleasant phenol odour, vapour volatility and sublimation. The compound darkens on exposure to air or light and evaporates with water vapour. Soluble in ethanol, ether, benzene, chloroform and alkali solutions, insoluble in water. In contact with ferric chloride produces a purple precipitate.
1-Naphthol is used as a precursor in the manufacturing of various azo dyes and pharmaceuticals such as nadolol. It is used as biomarkers. It is used in analytical chemistry as Molisch's reagent (1-naphthol dissolved in ethanol) for checking the presence of carbohydrates. It plays an essential role with sodium hypobromite to detect the presence of arginine in proteins, which is called as Sakaguchi test.
2-Isopropylnaphthalene can be used to synthesize 1-naphthol by oxidation via the hydroperoxide (Hock synthesis).
Historically 1-naphthol was produced via caustic fusion of naphthalene-1-sulfonic acid, but this was superseded by the I.G. Farbenindustrie process involving hydrolysis of 1-naphthylamine with aqueous 22 % sulfuric acid at 200°C under pressure in a lead-lined autoclave. To obtain a purer product, Union Carbide developed a process based on catalytic oxidation of tetralin to 1-tetralol and 1-tetralone followed by dehydrogenation. The two-stage catalytic process is claimed to give an overall yield of 72% 1-naphthol, with an overall efficiency of 97%.
1-Naphthol is a hydroxyl-aromatic compound. It has been used as a prooxidant to analyze its ability to induce hemolysis in a zebrafish G6PD (glucose-6-phosphate dehydrogenase) deficiency model.
ChEBI: 1-naphthol is a naphthol carrying a hydroxy group at position 1. It has a role as a genotoxin and a human xenobiotic metabolite.
1-Naphthol is reduced by sodium in liquid ammonia to give 5,6,7,8-tetrahydro-1-naphthol; oxidised by NaOCl - FeCl3 or I2-KI to give a purple colour. Phosphorus pentachloride is chlorinated at 150 °C to give 1-chloronaphthalene, thionyl chloride to give 4-chloro-1-naphthol, or Cl2-CH3COOH to give 2,4-dichloro-1-naphthol. Similarly, bromine produces 2,4-dibromo-1-naphthol, a reaction that can be used in quantitative titrations. Its nitration reaction produces a complex mixture. The crude 2,4-dinitro derivative is used as an acidic yellow dye, comparable to picric acid. Sulfonation at 50 °C readily produced 1-naphthol-2,4-disulfonic acid.
1-Naphthol, a metabolite of carbaryl and naphthanlene. It is formed by spontaneous reaction from (1R, 2S)-Naphthalene epoxide followed to form 1, 4-Dihydroxynaphthalene.
Toxic by ingestion and skin absorption.
Flammability and Explosibility
Not classified
1-Naphthol can be used in dye manufacture and is
classified as a hair dye. Combined with epichlorhydrin
and NaOH to form alpha-naphthyl glycidyl ether, it caused
sensitization in one of three workers in a chemical plant.
Poison by ingestion and
intraperitoneal routes. Moderately toxic by
skin contact. An experimental teratogen.
Experimental reproductive effects. A severe
eye and skin irritant. Mutation data reported.
Ingestion of large amounts can cause
nephritis, vomiting, diarrhea, circulatory
collapse, anemia, convulsions, and death.
Can cause kidney irritation and injury to
cornea and lens of the eye. Combustible
when exposed to heat or flame. When
heated to decomposition it emits acrid
smoke and irritating fumes.
To a 10 mL pressure vessel, aryl halide (1.00 mmol), copper (I) oxide (0.05 mol), ligand (0.05 mol), 3 M sodium hydroxide (2 mL), and organic solvent (2 mL) were added. The reaction mixture was irradiated at 160 °C for 10 min with strong stirring. The reaction mixture was allowed to cool to room temperature. The reaction mixture was filtered through a plug of celite in a fritted filter funnel and washed with ethyl acetate. The product was extracted using 30 mL of ethyl acetate for three times. The organic extract is washed three times with 10 mL water and two times with 10 mL of brine. The combined organic phases were dried over anhydrous MgSO4 and the solvent was removed under reduced pressure to afford 1-naphthol.
Sublime 1-naphthol, then crystallise it from aqueous MeOH (charcoal), aqueous 25% or 50% EtOH, *C6H6, cyclohexane, heptane, CCl4 or H2O. Dry it over P2O5 in vacuo. The 4-nitrobenzoate has m 143o (from EtOH). [Shizuka et al. J Am Chem Soc 107 7816 1985, Beilstein 8 H 596, 6 IV 4208.]