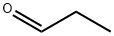Propionaldehyde is a volatile liquid substance which consists of one carbonyl group and its characteristic functional group. The main functional group is an aldehyde which classifies propionaldehyde in the carbonyls. The main trunk of this substance is a short aliphatic chain. The carbonyl group determines, to a large extent, its chemical properties and most importantly its nucleophilic property. Propionaldehyde is readily oxidized if in contact to oxygen and should therefore be stored under inert gases.
Propionaldehyde has a characteristic sharp and pungent odor similar to acetaldehyde.It undergoes reactions typical for the low molecular weight aldehydes, which, because of the terminal carbonyl group, are very reactive. Contamination or the exposure to elevated temperatures may induce a hazardous polymerization.

Propionaldehyde is a colorless, flammable liquid with a suffocating fruity odor. It is used as Intermediate for the chemical industry, for example for the manufacture of pharmaceuticals, pesticides, perfumes and plastics.
Reported found in apple aroma and in the essential oils of camphor, Rosa centifolia, clary sage, Pinus excelsa
and Pinus silverstris. Also reported found in over 100 natural products including apple, banana, sweet cherry, sour cherry, black
currant grapes, melon, pineapple, strawberry, cabbage, carrot, celery, cucumber, garlic, onion, leek, potato, peas, rutabaga, tomato,
Scotch spearmint oil, vinegar, bread and bread preferment, blue cheeses, cheddar cheese, Swiss cheese, butter, milk, cream, boiled
egg, caviar, fatty fish, cooked turkey, beef, pork and chicken, beer, rum, cognac, whiskies, grape wines, coffee, cocoa, tea, roasted
filberts and peanuts, peanut oil, potato chips, pecans, oats, honey, soybeans, Arctic bramble, beans, Bantu beer, plum brandy, cauliflower,
Brussels sprouts, rice, prickly pear, peated malt, clary sage, truffle, krill, oysters, loganberry, Chinese quince and maté.
Propionaldehyde is produced by the oxo reaction of ethylene with carbon monoxide and hydrogen. n-Propyl alcohol is produced by hydrogenation of propionaldehyde, and propionic acid is made by oxidation of propionaldehyde.
n-Propyl alcohol is used as solvent in printing inks and as an intermediate in the preparation of agricultural chemicals. Propionic acid is used as a grain preservative as, for example, in preventing spoilage of wet corn used as animal feed. The use of propionic acid as a grain preservative is an alterna tive to drying by heating, which consumes fuel, and is considered mostly when fuel is expensive.
Manufacture of propionic acid, polyvinyl, and
other plastics; synthesis of rubber chemicals; disin-
fectant; preservative.
Propionaldehyde is used in the productionof propionic acid, propionic anhydride, andmany other compounds. It is formed in theoxidative deterioration of corn products, suchas corn chips. It occurs in automobile exhaustgases.
By oxidation of propyl alcohol, or by dry distillation of barium propionate with calcium formate.
Propionaldehyde is prepared in the usual way by the oxidation of n-propyl alcohol. It can also be prepared by dehydrogenating the alcohol by passing the vapors over a heated copper or brass catalyst. This avoids the danger of further oxidation to propionic acid.
The reactions of propionaldehyde are practically like those of acetaldehyde. It must be remembered that only the two alpha hydrogen atoms are active in replacement and condensation reactions.
ChEBI: An aldehyde that consists of ethane bearing a formyl substituent. The parent of the class of propanals.
A clear colorless liquid with an overpowering fruity-like odor. Less dense than water. Flash point 15°F. Vapors are heavier than air.
Highly flammable. Soluble in water.
Propionaldehyde may form explosive peroxides. Reacts vigorously with oxidizing agents. Explosive in the form of vapor when exposed to heat or flame [Lewis]. Incompatible with strong bases and strong reducing agents. Vigorous polymerization reaction with methyl methacrylate. Polymerization may also occur in the presence of acids or caustics .
Flammable, dangerous fire risk, explosive
limits in air 3.0–16%. Upper respiratory tract irri-
tant.
Propionaldehyde is a mild irritant to humanskin and eyes. The irritation effect from40 mg was severe in rabbits’ eyes. The toxicityof this compound observed in test animalswas low. Subcutaneous administration in ratsexhibited the symptoms of general anestheticeffect, convulsion, and seizure. Inhalationtoxicity was determined to be low. A concentrationof 8000 ppm (19,000 mg/m3) inair was lethal to rats.
LD50 value, oral (rats): 1400 mg/kg
LD50 subcutaneous (rats): 820 mg/kg.
Behavior in Fire: Vapor is heavier than air and may travel considerable distance to a source of ignition and flash back.
Flammability and Explosibility
Flammable
Moderately toxic by
skin contact, ingestion, and subcutaneous
routes. Mddly toxic by inhalation. A skin
and severe eye irritant. Flammable liquid.
Dangerous fire hazard when exposed to heat
or flame; reacts vigorously with oxidizers.
Explosive in the form of vapor when
exposed to heat or flame. Vigorous
polymerization reaction with methyl
methacrylate. To fight fire, use alcohol
foam, CO2, dry chemical. When heated to
decomposition it emits acrid smoke and
irritating fumes. See also ALDEHYDES.
Used as a synthetic flavoring; as
a disinfectant and preservative; to make propionic acid;
in plastic and rubber manufacturing; to make alkyl resins
and plasticizers.
In a mutagenic test in V79 cells, Eder et al. observed
that propionaldehyde is not mutagenic at 1 mM, but is toxic at 2mM. Similar to acrolein, Eder et al. suggested that
the mutagenicity of this compound is mediated by its bifunctionality,
whereas its cytotoxicity is mediated by the aldehyde
function.
UN1275 Propionaldehyde, Hazard Class: 3; Labels: 3-Flammable liquid.
Propionaldehyde is readily oxidized if in contact to oxygen and should therefore be stored under inert gases.
the aldehyde with CaSO4 or CaCl2, and fractionally distil it under nitrogen or in the presence of a trace of hydroquinone (to retard oxidation). Blacet and Pitts [J Am Chem Soc 74 3382 1952] repeatedly distilled the middle fraction in a vacuum until it no longer gave a solid polymer when cooled to -80o. It is stored with CaSO4. [Beilstein 1 IV 3165.]
Incompatible with strong acids; amines.
Incompatible with oxidizers (chlorates, nitrates, peroxides,
permanganates, perchlorates, chlorine, bromine, fluorine,
etc.); contact may cause fires or explosions. Keep away
from alkaline materials, strong bases, strong acids, oxoa-
cids, epoxides. Strong caustics; reducing agents can cause
explosive polymerization. Can self-ignite if finely dispersed
on porous or combustible material, such as fabric. Heat or
UV light can cause decomposition. Aldehydes are fre-
quently involved in self-condensation or polymerization
reactions. These reactions are exothermic; they are often
catalyzed by acid. Aldehydes are readily oxidized to give
carboxylic acids. Flammable and/or toxic gases are gener-
ated by the combination of aldehydes with azo, diazo com-
pounds, dithiocarbamates, nitrides, and strong reducing
agents. Aldehydes can react with air to give first peroxo
acids, and ultimately carboxylic acids. These autoxidation
reactions are activated by light, catalyzed by salts of
transition metals, and are autocatalytic (catalyzed by the
products of the reaction). The addition of stabilizers (anti-
oxidants) to shipments of aldehydes retards autoxidation
The initial threshold screening level (ITSL) for propionaldehyde is 8 μg/m3 based on an annual averaging time.
Propionaldehyde is destroyed by burning in achemical incinerator equipped with an afterburnerand scrubber. Permanganate oxidationis a suitable laboratory method of destruction.