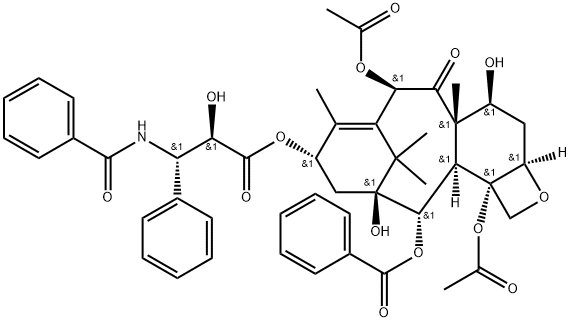
パクリタキセル
化学名:パクリタキセル
CAS番号.33069-62-4
英語名:Paclitaxel
CBNumberCB3273425
MFC47H51NO14
MW853.92
MOL File33069-62-4.mol
别名
パクリタキセル
タキソール
パクリタキセル (JAN)
more
パクリタキセル物理性質
| 融点 | 213 °C (dec.)(lit.) |
| 沸点 | 774.66°C (rough estimate) |
| 比旋光度 | D20 -49° (methanol) |
| 比重(密度) | 0.200 |
| 屈折率 | -49 ° (C=1, MeOH) |
| 闪点 | 9℃ |
| 貯蔵温度 | 2-8°C |
| 溶解性 | メタノール: 50 mg/mL、透明、無色 |
| 外見 | 粉 |
| 酸解離定数(Pka) | 11.90±0.20(Predicted) |
| 色 | 白い |
| 水溶解度 | 0.3mg/L(37℃) |
| 極大吸収波長 (λmax) | 227nm(MeOH)(lit.) |
| Merck | 14,6982 |
| BRN | 1420457 |
| 安定性: | 安定。強力な酸化剤とは相容れない。可燃性。 |
| InChIKey | RCINICONZNJXQF-MZXODVADSA-N |
| SMILES | N([C@H]([C@@H](O)C(=O)O[C@H]3C[C@]4([C@H](C6[C@]7([C@H](OC7)C[C@@H]([C@]6(C(=O)[C@@H](C(=C3C)C4(C)C)OC(=O)C)C)O)OC(=O)C)OC(=O)c5ccccc5)O)c2ccccc2)C(=O)c1ccccc1 |
show more
| 主な危険性 | Xn |
| Rフレーズ | 37/38-41-42/43-62-68-40-48-20/21/22-68/20/21/22 |
| Sフレーズ | 22-26-36/37/39-45 |
| RIDADR | 1544 |
| WGK Germany | 3 |
| RTECS 番号 | DA8340700 |
| F | 10-21 |
| 国連危険物分類 | 6.1(b) |
| 容器等級 | III |
| HSコード | 29329990 |
| ストレージクラス | 6.1C - Combustible, acute toxic Cat.3 toxic compounds or compounds which causing chronic effects |
| Hazard Classifications | Muta. 2 Repr. 1B STOT RE 2 |
| 有毒物質データの | 33069-62-4(Hazardous Substances Data) |
| 毒性 | LD50 intraperitoneal in mouse: 128mg/kg |


