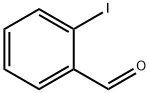
Chemical Properties
white to light beige low melting crystalline
Uses
2-Iodobenzaldehyde may be used as a reactant in the synthesis of the following heterocycles:
- 2,3-diaryl-1-indenones
- indolo[1,2-a]quinazolines
- Baylis-Hillman (BH) adducts
It may also be used in preparing:
- 5-phenylindazolo[3,2-b]quinazolin-7(5H)-one
- 4-(3-iodophenyl)-2,2:6,2-terpyridine
- fluoren-9-one
- 2-formyl-3′-methoxybiphenyl
Uses
2-Iodo-benzaldehyde is a very useful synthetic intermediate. It is halogenated Benzaldehyde (B119740) which is mainly used as a precursor to other organic compounds, such as pharmaceuticals, and plastic additives.
General Description
2-Iodobenzaldehyde (
o-iodobenzaldehyde) is a 2-halobenzaldehyde derivative. Its crystals belong to the orthorhombic crystal system and
P2
12
12
1 space group.
Synthesis
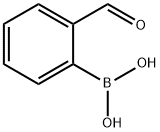
General procedure for the synthesis of 2-iodobenzaldehyde from 2-formylphenylboronic acid: 2-formylphenylboronic acid (0.067 g, 0.4 mmol), copper powder (0.0052 g, 0.08 mmol), (CF3)2CFI (0.178 g, 0.6 mmol) and DMF (2 mL) were added to a closed tube with a rubber stopper. The reaction mixture was stirred at room temperature for 24 hours. Upon completion of the reaction, the resulting suspension was poured into water and extracted with ethyl acetate. The organic layers were combined, dried with anhydrous Na2SO4 and concentrated under reduced pressure to remove the solvent. The crude product was purified by silica gel column chromatography using petroleum ether/ethyl acetate (20:1, v/v) as eluent to give 0.086 g of 2-iodobenzaldehyde as a light yellow solid (0.35 mmol, 87% yield).
References
[1] Journal of Fluorine Chemistry, 2016, vol. 189, p. 59 - 67