Synthesis
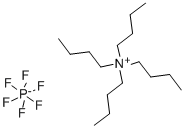
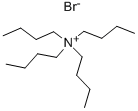
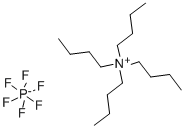
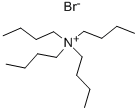
The general procedure for the synthesis of tetrabutylammonium hexafluorophosphate from tetrabutylammonium bromide was as follows: potassium hexafluorophosphate (K[PF6], 2.055 g, 11.2 mmol) was dissolved in 30 mL of deionized water. Tetrabutylammonium bromide ([(n-Bu4)N]Br, 3.601 g, 11.2 mmol) was dissolved in 30 mL of dichloromethane (CH2Cl2) and the resulting solution was subsequently added to the aqueous potassium hexafluorophosphate solution. After stirring the reaction mixture for 24 h at room temperature, phase separation was carried out. The organic phase was washed three times with 10 mL of deionized water, followed by drying with anhydrous sodium carbonate (Na2CO3) and filtration. The filtrate was concentrated by rotary evaporation to give a white solid product. The resulting solid was dried under vacuum at 80 °C for 18 h to give 3.822 g of tetrabutylammonium hexafluorophosphate ([(n-Bu4)N][PF6]) in 88% yield (9.87 mmol). The product characterization data are as follows: differential scanning calorimetry (DSC, 10 K/min): melting point (m.p.) = 250 °C, decomposition temperature (Tdec) = 388 °C. Calculated elemental analysis (measured values): C 49.60% (49.97%), H 9.37% (9.00%), N 3.61% (3.61%).1H NMR (25 °C, CD3CN, 300.13 MHz, δ ppm): 0.96 (t, 12H, CH3), 1.34 (m, 8H, CH3-CH2), 1.59 (m , 8H, CH2-CH2N), 3.06 (m, 8H, NCH2).19F NMR (25 °C, CD3CN, 300.13 MHz, δ ppm): -73.0 (d, 6F, PF6, JP-F = 706.7 Hz).31P NMR (25 °C, CD3CN, 300.13 MHz, δ ppm): - 144.6 (sept, 1P, PF6, JP-F = 706.7 Hz). Infrared spectrum (ATR, 32 scans, ν cm-1): 2966 (m), 2937 (w), 2879 (w), 1472 (m), 1404 (w), 1386 (w), 1360 (w), 1350 (w), 1319 (w), 1260 (w), 1242 (w), 1165 (w), 1109 (w), 1070 (w), 1035 (w), 931 (w), 880 (m), 829 (s), 738 (m), 555 (s).
References
[1] European Journal of Organic Chemistry, 2012, # 19, p. 3566 - 3569
[2] Patent: WO2014/167034, 2014, A2. Location in patent: Page/Page column 48; 49
[3] Angewandte Chemie - International Edition, 2015, vol. 54, # 15, p. 4474 - 4477
[4] Angew. Chem., 2015, vol. 127, # 15, p. 4556 - 4559,4
[5] Chemistry - A European Journal, 2016, vol. 22, # 12, p. 4175 - 4188