Synthesis
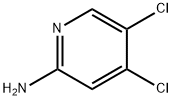
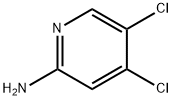
General procedure for the synthesis of 4,5-dichloropyridin-2-amine from 2-amino-4-chloropyridine: N-chlorosuccinimide (NCS, 2.67 g, 20.0 mmol) was added to a solution of 2-amino-4-chloropyridine (1.28 g, 10.0 mmol) in DMF (40 mL) at -20 °C. The reaction mixture was gradually warmed to room temperature and stirred continuously for 24 hours. Upon completion of the reaction, the mixture was poured into ice water (300 mL) and extracted with ethyl acetate. The combined organic phases were washed sequentially with 1 M sodium hydroxide solution and saturated saline, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by silica gel column chromatography to afford the target compound 4,5-dichloropyridin-2-amine (1.12 g, 69.0% yield).
References
[1] Bioorganic and Medicinal Chemistry Letters, 2016, vol. 26, # 1, p. 60 - 67
[2] European Journal of Medicinal Chemistry, 2016, vol. 117, p. 19 - 32
[3] Patent: WO2016/124553, 2016, A1. Location in patent: Page/Page column 71-72
[4] Patent: WO2018/11138, 2018, A1. Location in patent: Page/Page column 69-70
[5] Synthetic Communications, 1997, vol. 27, # 5, p. 861 - 870