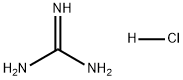
description
Guanidine Hydrochloride is the hydrochloride salt form of guanidine, a strong basic compound with parasympathomimetic activity. Guanidine hydrochloride enhances the release of acetylcholine following a nerve impulse and potentiates acetylcholine actions on muscarinic and nicotinic receptors. It also appears to slow the rates of depolarization and repolarization of muscle cell membranes.
Guanidine Hydrochloride is a potent chaotropic agent extensively employed for protein and nucleic acid purification. It can denature and refold proteins, isolate RNA, and recover periplasmic proteins. Even insoluble, denatured proteins, such as inclusion bodies, can be solubilized with this powerful denaturant at high concentrations. At lower concentrations, Guanidine Hydrochloride can facilitate the refolding of denatured proteins and the restoration of enzymatic activity. Additionally, Guanidine Hydrochloride is known to inhibit RNase.
Protein Denaturation
Guanidine Hydrochloride (GdnHCl) is a better denaturant than urea.It should normally give higher and lower values for m and for Cm, respectively, but identical deltaG° values. Since m values in both denaturants can be predicted/estimated from the size of the protein (Myers et al., 1995) and delatG° should not change, yes it is possible-in principle-to predict Cm in one denaturant, knowing its value in the other.
but to have a general correlation co-efficient is almost impossible as every protein has unique folding. Though such co-relation may be found for a special protein fold, but what purpose it may serve apart from giving a tentative idea that GdnHCl or urea may be a better denaturant! If you are looking for even better denaturant than Gdn HCl then use GdnSCN, it is better and more effective
Uses
Guanidine hydrochloride is used in RNA isolation to dissociate nucleoproteins and inhibit RNase.
Strong chaotropic agent useful for the denaturation and subsequent refolding of proteins. This strong denaturant can solubilize insoluble or denatured proteins such as inclusion bodies. This can be used as the first step in refolding proteins or enzymes into their active form. Urea and dithiothreitol (DTT) may also be necessary.
Drug intermediates
Guanidine hydrochloride drug used primarily as an intermediate in the manufacture of sulfadiazine, an important raw material sulfamethyldiazine, sulfamethazine and folic acid.
Guanidine hydrochloride (or guanidine nitrate) and ethyl cyanoacetate reaction, cyclization of 2,4-diamino-6-hydroxy pyrimidine, folic acid for the synthesis of antianemic. Furthermore guanidine hydrochloride also used synthetic anti-static agent.
Preparation
In Dicyanodiamide and ammonium (Ammonium chloride) as raw material, melt reaction at 170-230 ℃, guanidine hydrochloride obtained crude, refined products.
Reference quality standards
Name of Project Specifications
Pharmaceutical grade industrial grade
Appearance white crystal; white crystal
Content > 99% 99.5%
Ammonium <0.3% 0.5%
Water Insoluble substance <0.1% 0.1%
Moisture <0.3% 0.3%
Ash <0.05% 0.05%
Water-soluble test qualified; qualified
Melting range °C 184-188; 182-188
Absorbance (UV wavelength 260NM) <=0.04
Absorbance (UV wavelength 280NM) <=0.02
PH value (4% aqueous solution) 6.4 ± 0.2 6.4 ± 0.2
Chemical Properties
Guanidine hydrochloride, a strong organic base, appears as white crystal powder or yellowish lumps that are almost insoluble in acetone, benzene, and ether. In physiological pH, it exists primarily as guanidium ions. It is a natural byproduct of protein metabolism found in urine and is commonly used in protein denaturation in laboratory research.
Uses
Guanidine has a good bacterial and fungicidal efficacy when used in low concentration and can also be used as disinfectant.
Uses
Guanidine hydrochloride is used for purification of proteins and nucleic acids. It can be used as a medicine, organic synthesis intermediate and is used in dyes. It acts as an intermediate in the preparation of sulfadiazine, which is an important raw material for sulfamethyldiazine, sulfamethazine and folic acid. It is utilized in the synthesis of 2-amino-pyrimidine, 2- amino -6- methyl-pyrimidine and 2-amino -4,6 - dimethyl-pyrimidine. Also used in RNA isolation to dissociate nucleoproteins and inhibit RNase.
Application
Guanidine hydrochloride can be used as pharmaceuticals, pesticides, dyes and other organic synthesis intermediates. It can be used to synthesize 2-aminopyrimidine, 2-amino-6-methylpyrimidine, 2-amino-4,6-dimethylpyrimidine, and is used for the manufacture of sulfadiazine, sulfamethazine, sulfamethazine and other sulfa drugs. Intermediate. Guanidine hydrochloride (or guanidine nitrate) reacts with ethyl cyanoacetate to form 2,4-diamino-6-hydroxypyrimidine, which is used to synthesize the anti-anemia drug folic acid. It can also be used as an antistatic agent for synthetic fibers. Can also be used for protein denaturants. As a strong denaturant in experiments for extracting total cellular RNA[3]. The guanidine hydrochloride solution can dissolve the protein, leading to the destruction of the cell structure, the destruction of the secondary structure of the nucleoprotein, and the dissociation from the nucleic acid. In addition, the RNase can be inactivated by reducing agents such as guanidine hydrochloride.
Definition
ChEBI: Guanidine Hydrochloride is an aminocarboxamidine, the parent compound of the guanidines. It is a strong organic base existing primarily as guanidium ions at physiological pH. It is found in the urine as a normal product of protein metabolism.
Preparation
The process for preparing guanidine hydrochloride is as follows:
Step 1: Fritting - Combine dicyandiamide and ammonium chloride in a reactor with a 1:1.27 weight ratio and carry out a frit reaction at 170-230°C to obtain crude guanidine hydrochloride.
Step 2: Dissolving - Dissolve the crude guanidine hydrochloride in water at a 1:1 ratio at normal temperature. Then, add ammonium salt alkaloid in an amount determined by the ammonium salt content in the crude guanidine hydrochloride.
Step 3: Filtering - Remove raw materials and reaction by-products with filtration.
Step 4: Dehydrating - Dehydrate the filtered ground mother liquor at a high temperature.
Step 5: Crystallizing - Cool the supersaturated solution, concentrate, and crystallize to obtain high-purity guanidine hydrochloride.
Step 6: Repeat steps 2-5 and add dicyandiamide in the reactor with the guanidine hydrochloride to carry out frit reaction at 170-230°C for 3-4 hours to obtain even higher purity guanidine hydrochloride. This step can be repeated several times if needed.
Step 7: To obtain higher purity guanidine hydrochloride, subject the guanidine hydrochloride obtained in step 5 to steps 2-5 again, with the water ratio in step 2 adjusted to 1.5:1. This step can be repeated numerous times as required.
General Description
Guanidine hydrochloride (GdnHCl) is a well-known protein and enzyme denaturant. It is a small molecule and hydroscopic in nature. It is used to synthesis medicine, pesticide, dye and other organic compound, is the important material to make sulfanilamide medicine and folacin, and can be used as anti-static agent of compound fibre.
Hazard
Toxic by ingestion, evolves hydrogen chloride
fumes on heating.
Biochem/physiol Actions
Guanidine hydrochloride (GdnHCl) is involved in conformational changes and loss of enzyme activity due to inactivation of the enzyme. It is involved in the loss of activity of alkaline phosphatase (ALPase) enzyme in Haliotis diversicolor. GdnHCl functions by hampering the activity of heat shock protein 104 (Hsp104) adenosine triphosphatase (ATPase) in vivo. It can also inactivate enzymes like papain and aminocyclase. Higher concentrations of GdnHCl leads to viral inactivation of herpes simplex virus 1 (HSV-1).
Purification Methods
Crystallise the hydrochloride from hot methanol by chilling to about -10o, with vigorous stirring. The fine crystals are filtered through fritted glass, washed with cold (-10o) methanol, dried at 50o under vacuum for 5hours. (The product is purer than that obtained by crystallisation at room temperature from methanol by adding large amounts of diethyl ether.) [Kolthoff et al. J Am Chem Soc 79 5102 1957, Beilstein 3 H 86, 3 II 71, 3 III 160, 3 IV 150.]