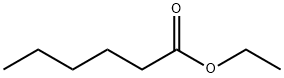
Description
Ethyl caproate (also ethyl hexanoate) is naturally found in the fruits of Ananas sativus. It can be synthesized by the direct esterification of caproic acid with ethyl alcohol. It has strong, sweet-ethereal like pineapple odor, with nuances of banana and strawberry.
Ethyl caproate is approved by the FDA for food use (as a flavoring agent in desserts and beverages) without hazard to public health. Ethyl Caproate is used to synthesize novel EP2/EP4 dual agonist of γ-lactam prostaglandin E1 analogs. It is also used as a chemical reagent in the synthesis of PPARα antagonists in the treatment of metabolic diseases.
References
[1] D. L. J. Opdyke (1974) Monographs on Fragrance Raw Materials
[2] https://www.trc-canada.com
Chemical Properties
Ethyl hexanoate has a powerful, fruity odor with a pineapple–banana note. It has been also reported to have a winy odor.
Chemical Properties
CLEAR COLOURLESS LIQUID
Chemical Properties
Ethyl Hexanoate is a colorless liquid
with a strong fruity odor, reminiscent of pineapples. It occurs in many fruits and
is used in small amounts for floral, fruity notes in perfume compositions and in
larger quantities in fruit flavors.
Occurrence
Reported found in strawberry, rum, bourbon, cocoa, kiwi fruit, black currant, apple, orange and grapefruit
juice, guava, Vitis vinifera, pineapple, strawberry jam, clove bud, cheeses, cognac, whiskies, grape wines, passion fruit juice, mango,
fruit brandies, figs, corn oil, mountain papaya, pawpaw and mastic gum leaf oil.
Uses
Ethyl Caproate is used in the synthesis of novel EP2/EP4 dual agonist of γ-lactam prostaglandin E1 analogs. Also used as a chemical reagent in the synthesis of PPARα antagonists in the treatment of metabolic diseases.
Uses
Ethyl hexanoate may be used as an analytical reference standard for the determination of the analyte in wine and beer samples by chromatography based techniques.
Uses
manufacture of artificial fruit flavors.
Definition
ChEBI: A fatty acid ethyl ester obtained by the formal condensation of hexanoic acid with ethanol.
Preparation
By esterification of caproic acid with ethyl alcohol in the presence of concentrated H2SO4 or HCl
Aroma threshold values
Detection: 0.3 to 5 ppb
Taste threshold values
Taste characteristics at 10 ppm: fruity and waxy with a tropical nuance.
General Description
Ethyl hexanoate is one of the odorants contributing to the typical guava aroma. It also contributes to the fresh strawberry aroma.
Safety Profile
A skin irritant.
Flammable liquid when exposed to heat or
flame; can react with oxidzing materials.
When heated to decomposition it emits
acrid smoke and irritating fumes. To fight
fire, use CO2, foam, dry chemical. See also
ESTERS.
Carcinogenicity
Not listed by ACGIH, California
Proposition 65, IARC, NTP, or OSHA.
Metabolism
Aliphatic esters, including ethyl caproate, are thought to be readily hydrolysed to the corresponding alcohol and acid, which are then further metabolized (Fassett, 1963). Ethyl caproate administered orally to rats produced a uniform ketonuria and it was considered probable that caproic acid was broken down chiefly by ?-oxidation (Deuel, Hallman, Butts & Murray, 1936). When 2 g ethyl caproate dissolved in aqueous ethanol was fed directly into the rumen of a cow, 0.003°/ was transferred to the milk, reaching a maximum level of 60 /fg/litre after 2-4 hr (Honkanen, Karvonen & Virtanen, 1964). The energy from ethyl caproate was 52% available when the ester was fed to four chicks at a level of 5% in the diet (Yoshida et al. 1970).
Solubility in organics
Soluble in oils and alcohol. Poorly soluble in Ropylene glycol, insoluble in Glycerin.