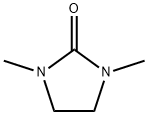
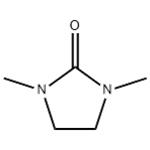
DMI, a high purity aprotic solvent, is composed of nitrogen containing five membered ring compound and widely recognized as an epoch making solvent. It is a colorless, transparent, high polar solvent with high thermal and chemical stability and non-corrosiveness. The material has a high boiling point of 222° C, a high flash point of 120° C (open method) / 95° C (close method), and a low melting point of 7.5° C.
Almost colorless, clear liquid
1,3-Dimethyl-2-imidazolidinone may be used:
- As substitute solvent for hexamethylphosphoric triamide (HMPA) in the synthesis of 1,2-bis(trimethylsilyl)benzene.
- As solvent during the α-regioselective prenylation of imine.
- As component of mobile phase for the size-exclusion chromatographic analysis of cellulose.
1,3-Dimethyl-2-imidazolidinone is used as a high-boiling polar aprotic solvent. It is used as a substitute solvent for toxic hexamethylphosphoramide. It finds application in detergents, dyestuffs, electronic materials and in the manufacture of polymers. It is involved in the preparation of 1,2-bis(trimethylsilyl)benzene. Further, it is utilized as a solvent during alfa-regioselective prenylation of imines. In addition to this, it constitutes the mobile phase during size-exclusion chromatographic analysis of cellulose.
DMI can be used in a variety of applications including detergents, dyestuffs, electronic materials and in the manufacture of polymers. Its versatility can be attributed to it. Of particular note are its excellent solubility for inorganic and organic compounds and high dielectric constant and solvation effect.
ChEBI: 1,3-Dimethyl-2-imidazolidinon is an imidazolidinone.
1,3-Dimethyl-2-imidazolidinone (DMI) is an imidazolidine derivative. It is reported to act as a promoter by minimizing the formation of dialkylation byproducts and accelerating the rate of monoalkylation of γ-butyrolactone. Conversion of CO2 to form lower alcohols by homogeneous catalytic hydrogenation using DMI as solvent has been investigated.
Flammability and Explosibility
Non flammable
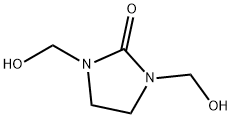
Example 4: 320 g (2.2 mol) of 1,3-bis(hydroxymethyl)imidazolidin-2-one and 200 g (2.9 mol) of sodium formate were dissolved in 620 g (13.5 mol) of formic acid, and the reaction mixture was heated under reflux conditions for 8 hours. After completion of the reaction, 238 g of formic acid was removed by distillation under reduced pressure. To the residue 200 g of potassium carbonate was slowly added. After cooling, the crude product was separated by decantation and washed with a small amount of dichloromethane. Subsequently, the product was purified by vacuum distillation. 157 g of 1,3-dimethyl-2-imidazolidinone was collected in 63% yield at 90°C to 95°C and a pressure of 3 torr. The purity of the product was determined by gas chromatography (GC) and hydrogen nuclear magnetic resonance (1H-NMR) and confirmed to be >98%.
Due to a lack of available toxicity data the screening level is being set at default value of 0.1 μg/m3 with annual averaging.
[1] Patent: US4864026, 1989, A