General procedure for the synthesis of 3,5-difluorochlorobenzene from 6-chloro-2,4-difluoroaniline:
1. 0.5 moles (1.0 equiv.) of 6-chloro-2,4-difluoroaniline and 200 ml of dichloromethane were added to a reaction flask and stirred until completely dissolved.
2. 0.55 moles (1.1 eq.) of N-chlorosuccinimide (NCS) was added in 5 batches, the reaction temperature was controlled at 10~15°C, and after the addition was completed, the reaction was heated to reflux for 2 hours.
3. The completion of the reaction was monitored by TLC and the pH was adjusted to 6-7 by adding 10% dilute hydrochloric acid to the reaction mixture.
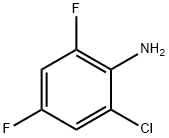
4. The organic phase was separated, washed with water and rotary evaporated to dryness to give 0.44 mol (0.88 eq.) of 2,4-difluoro-5-chloroaniline.
5. 0.44 mol of 2,4-difluoro-5-chloroaniline was added to 1.5 mol (3 eq.) of 25% dilute sulfuric acid and stirred for 0.5 hr.
6. Add 0.75 mol (1.5 eq.) of sodium phosphite, control the reaction temperature at 30~35°C, slowly add 0.5 mol (1 eq.) of aqueous sodium nitrite dropwise, control the rate of dropwise acceleration so that the reaction temperature rises 4~5°C per hour.
7. Warm up to 60°C, keep the temperature stirring for 2 hours. 8.
8. leave to stratify, the organic phase is washed with water and then steam distilled. 9.
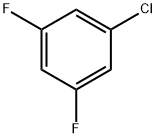
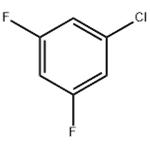
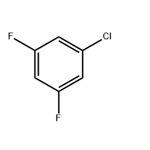
9. 0.41 moles (0.82 equivalents) of 3,5-difluorochlorobenzene were obtained by distillation, with a purity of 99.8%.