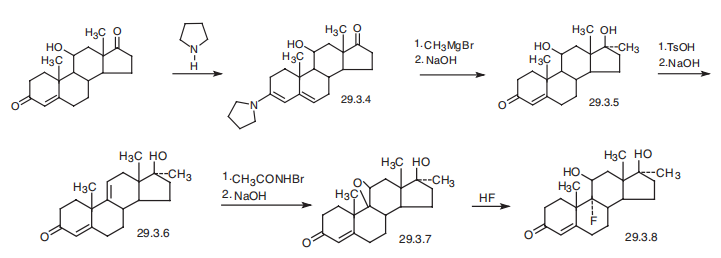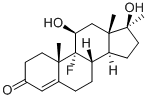
76-43-7
| Name | Fluoxymesterone |
| CAS | 76-43-7 |
| EINECS(EC#) | 200-961-8 |
| Molecular Formula | C20H29FO3 |
| MDL Number | MFCD00010480 |
| Molecular Weight | 336.44 |
| MOL File | 76-43-7.mol |
Chemical Properties
| Appearance | white to light yellow crystal powde |
| Melting point | 240 °C |
| alpha | 104 º (c=1,EtOH) |
| Boiling point | 474.2±45.0 °C(Predicted) |
| density | 1.0455 (estimate) |
| storage temp. | -20°C |
| solubility | H2O: ≤0.5 mg/mL |
| form | solid (photosensitive) |
| pka | 13.40±0.70(Predicted) |
| color | white |
| Water Solubility | NEGLIGIBLE |
| Merck | 13,4212 |
| InChI | InChI=1/C20H29FO3/c1-17-8-6-13(22)10-12(17)4-5-15-14-7-9-19(3,24)18(14,2)11-16(23)20(15,17)21/h10,14-16,23-24H,4-9,11H2,1-3H3/t14-,15-,16-,17-,18-,19-,20-/s3 |
| InChIKey | YLRFCQOZQXIBAB-MGWAJYFFNA-N |
| SMILES | [C@]12(F)[C@@H](O)C[C@]3(C)[C@](CC[C@@]3([H])[C@]1([H])CCC1=CC(=O)CC[C@]21C)(O)C |&1:0,2,5,7,10,12,22,r| |
| CAS DataBase Reference | 76-43-7(CAS DataBase Reference) |
| NIST Chemistry Reference | 4-Androsten-3-one, 9alpha-fluoro-11beta,17beta-dihydroxy-17alpha-methyl-,(76-43-7) |
| EPA Substance Registry System | 76-43-7(EPA Substance) |
Safety Data
| Hazard Codes | Xn |
| Risk Statements | |
| Safety Statements | |
| WGK Germany | 3 |
| RTECS | BV8390000 |
| HS Code | 29372900 |
| Hazardous Substances Data | 76-43-7(Hazardous Substances Data) |
| Toxicity |