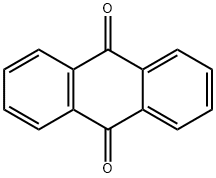
Антрахинон
- английское имяAnthraquinone
- CAS №84-65-1
- CBNumberCB5420557
- ФормулаC14H8O2
- мольный вес208.21
- EINECS201-549-0
- номер MDLMFCD00001188
- файл Mol84-65-1.mol
| Температура плавления | 284-286 °C (lit.) |
| Температура кипения | 379-381 °C (lit.) |
| плотность | 1.438 |
| плотность пара | 7.16 (vs air) |
| давление пара | 1 mm Hg ( 190 °C) |
| показатель преломления | 1.5681 (estimate) |
| Fp | 365 °F |
| температура хранения | no restrictions. |
| растворимость | 0.00007g/l |
| форма | Powder |
| цвет | Yellow-green to khaki to tan |
| Растворимость в воде | <0.1 g/100 mL at 23 ºC |
| Мерк | 14,687 |
| БРН | 390030 |
| Стабильность | Stable. Incompatible with strong oxidizing agents. Combustible. |
| ИнЧИКей | RZVHIXYEVGDQDX-UHFFFAOYSA-N |
| LogP | 3.39-3.4 at 30℃ |
| Непрямые добавки, используемые в веществах, контактирующих с пищевыми продуктами | 9,10-ANTHRAQUINONE |
| Справочник по базе данных CAS | 84-65-1(CAS DataBase Reference) |
| Рейтинг продуктов питания EWG | 2 |
| Словарь онкологических терминов NCI | anthracenedione; anthraquinone |
| FDA UNII | 030MS0JBDO |
| Предложение 65 Список | Anthraquinone |
| МАИР | 2B (Vol. 101) 2013 |
| Справочник по химии NIST | 9,10-Anthraquinone(84-65-1) |
| Система регистрации веществ EPA | Anthraquinone (84-65-1) |
| Коды опасности | Xi | |||||||||
| Заявления о рисках | 43-36/37/38 | |||||||||
| Заявления о безопасности | 36/37-37/39-26-24 | |||||||||
| РИДАДР | 3077 | |||||||||
| WGK Германия | 1 | |||||||||
| RTECS | CB4725000 | |||||||||
| Температура самовоспламенения | 650 °C | |||||||||
| TSCA | Yes | |||||||||
| кода HS | 2914 61 00 | |||||||||
| Класс опасности | 9 | |||||||||
| Группа упаковки | III | |||||||||
| Банк данных об опасных веществах | 84-65-1(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 orally in Rabbit: > 5000 mg/kg | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)


-
сигнальный язык
опасность
-
вредная бумага
H317:При контакте с кожей может вызывать аллергическую реакцию.
H350:Может вызывать раковые заболевания.
-
оператор предупредительных мер
P202:Перед использованием ознакомиться с инструкциями по технике безопасности.
P261:Избегать вдыхания пыли/ дыма/ газа/ тумана/ паров/ аэрозолей.
P272:Не уносить загрязненную спецодежду с места работы.
P280:Использовать перчатки/ средства защиты глаз/ лица.
P302+P352:ПРИ ПОПАДАНИИ НА КОЖУ: Промыть большим количеством воды.
P308+P313:ПРИ подозрении на возможность воздействия обратиться за медицинской помощью.
Антрахинон химические свойства, назначение, производство
Химические свойства
Anthraquinone is a combustible, light yellow to green crystalline solid. Soluble in ethanol, ether and acetone, insoluble in water. It may be prepared by reacting benzene with phthalic anhydride. The compoundis the basis of a range ofdyestuffs.Вхождение
Anthraquinones naturally occur in some plants (eg. aloe, senna, rhubarb, and Cascara buckthorn), fungi, lichens, and insects, where they serve as a basic skeleton for their pigments. Natural anthraquinone derivatives tend to have laxative effects.Использование
Anthraquinone is used in paper industry as a catalyst to increase the pulp production yield and to improves the fiber strength through reduction reaction of cellulose to carboxylic acid. It is also used as a precursor for dye formation.Определение
ChEBI: Anthraquinone is an anthraquinone that is anthracene in which positions 9 and 10 have been oxidised to carbonyls. It is a colorless crystalline quinone used in producing dyestuffs such as alizarin.Подготовка
Anthraquinone is obtained by oxidation of anthracene using sodium dichromate plus sulfuric acid, and is purified by dissolving in concentrated sulfuric acid at 130 °C and pouring into boiling water, whereupon anthraquinone separates as pure solid, and is recovered by filtration. Further purification may be accomplished by sublimation or crystallization from nitrobenzene, aniline or tetrachloroethane.Общее описание
Anthraquinone is a polycyclic aromatic hydrocarbon derived from anthracene or phthalic anhydride. Anthraquinone is used in the manufacture of dyes, in the textile and pulp industries, and as a bird repellant.Реакции воздуха и воды
Insoluble in water.Профиль реактивности
Anthraquinone is incompatible with strong oxidizing agents.Опасность
Possible carcinogen.Пожароопасность
Anthraquinone is combustible.Сельскохозяйственное использование
Repellent, Seed treatment: Used as a seed dressing or protectant. Banned in EU.Торговое название
(p)ANTHRAPEL®; FLIGHT CONTROL- PLUS®; HOELITE®; MORKIT®; REPELL®Фармаколо?гия
Anthraquinone is a secondary repellent and affects birds by causing post-ingestional distress (40). Sometimes, ingestion of anthraquinone-treated food produces vomiting, but often vomiting does not occur and the bird just sits quietly until the discomfort passes. Unlike methiocarb, anthraquinone doe not affect the bird’s nervous system and does not immobilize affected birds. Presumably, the emetic response is produced through irritation of the gut lining, but the actual mechanism is unclear. It is clear, however, that anthraquinone is not a taste repellent or contact irritant. Birds do not hesitate to eat treated food, and they exhibit no sign that treated food is unpalatable to them. The post-ingestional discomfort that results from eating anthraquinone-treated food produces a conditioned aversion to that food type. Birds need to experience the adverse consequences before learning to avoid the protected food. Thus, it is not reasonable to expect losses to cease immediately upon application of the repellent. There will be some level of loss in the crop as the depredating birds acquire the learned avoidance response.Профиль безопасности
Moderately toxic by intraperitoneal route. A mild allergen. Mutation data reported. Combustible when exposed to heat or flame. To fight fire, use water, foam, CO2, water spray or mist, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes.Возможный контакт
Anthraquinone is an important starting material for vat dye manufacture. Also used in making organics; and used as a bird repellent in seeds.Перевозки
UN3143 Dyes, solid, toxic, n.o.s. or Dye intermediates, solid, toxic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required.Методы очистки
Crystallise anthraquinone from CHCl3 (38mL/g), *benzene, or boiling acetic acid, wash it with a little EtOH and dry it under vacuum over P2O5. [Beilstein 7 IV 2556.]Несовместимости
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxidesУтилизация отходов
Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed.Антрахинон запасные части и сырье
сырьё
1of3
запасной предмет
- Anthrone
- 2,6-Diaminoanthraquinone
- 1 5-АНТРАХИНОНДИСУЛЬФОННАЯ КИСЛОТА
- Vat Green 3
- 1-Anthraquinonesulfonic acid
- 1,5-дихлорантрахинона
- 1-хлорантрахинон
- 9,10-диметилантрацен
- Водорода пероксид
- 1 5-DINITROANTHRAQUINONE 97
- Ализарин
- 2-антрахинонсульфоновая кислота
- Этилгексаноа
- 1,8-DINITROANTHRAQUINONE
- 1,4,5,8-Tetrachloroanthraquinone
1of5
Антрахинон поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия |
|---|---|---|---|---|
| +86-18400010335 +86-18034520335 |
China | 981 | 58 | |
| +8618629063126 | China | 346 | 58 | |
| +8613343047651 | China | 3002 | 58 | |
| +86-18032476855 +86-18032476855 |
China | 952 | 58 | |
| +8615373025980 | China | 870 | 58 | |
| 571-85586718 +8613336195806 |
China | 29798 | 60 | |
| +86-0371-55170693 +86-19937530512 |
China | 21670 | 55 | |
| +86-0551-65418679 +86-18949832763 |
China | 2989 | 55 | |
| 18017610038 | CHINA | 3620 | 58 | |
| +86-0371-86658258 +8613203830695 |
China | 29897 | 58 |