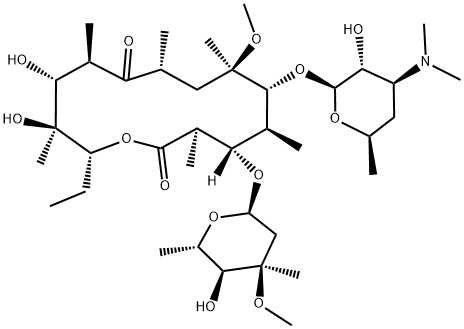
Кларитромицин
- английское имяClarithromycin
- CAS №81103-11-9
- CBNumberCB2225614
- ФормулаC38H69NO13
- мольный вес747.95
- EINECS1806241-263-5
- номер MDLMFCD00865140
- файл Mol81103-11-9.mol
химическое свойство
| Температура плавления | 217-220°C |
| Температура кипения | 86℃ |
| альфа | D24 -90.4° (c = 1 in CHCl3) |
| плотность | 1.1334 (rough estimate) |
| показатель преломления | -92 ° (C=1, CHCl3) |
| Fp | >110°(230°F) |
| температура хранения | -20°C |
| растворимость | Soluble in DMSO |
| пка | pKa 8.99(H2O t=25.0 I=0.167) (Uncertain) |
| форма | powder |
| цвет | colorless to faint yellow |
| Биологические источники | synthetic |
| Растворимость в воде | 99.48mg/L(20 ºC) |
| Мерк | 14,2339 |
| BCS Class | 4 |
| Стабильность | Store in Freezer |
| ИнЧИКей | AGOYDEPGAOXOCK-KCBOHYOISA-N |
| Рейтинг продуктов питания EWG | 1 |
| Словарь онкологических терминов NCI | clarithromycin |
| FDA UNII | H1250JIK0A |
| Словарь наркотиков NCI | Biaxin |
| Код УВД | J01FA09 |
| Предложение 65 Список | Clarithromycin |
| Система регистрации веществ EPA | Erythromycin, 6-O-methyl- (81103-11-9) |
| UNSPSC Code | 51102829 |
| NACRES | NA.85 |
больше
| Коды опасности | Xn,Xi | |||||||||
| Заявления о рисках | 22-36/37/38 | |||||||||
| Заявления о безопасности | 26-36-60 | |||||||||
| WGK Германия | 3 | |||||||||
| RTECS | KF4997000 | |||||||||
| кода HS | 29419000 | |||||||||
| Банк данных об опасных веществах | 81103-11-9(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 in male, female mice, male, female rats (mg/kg): 2740, 2700, 3470, 2700 orally, 1030, 850, 669, 753 i.p., >5000 all s.c. (Abe) | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)

-
сигнальный язык
предупреждение
-
вредная бумага
H302:Вредно при проглатывании.
-
оператор предупредительных мер
P264:После работы тщательно вымыть кожу.
P270:При использовании продукции не курить, не пить, не принимать пищу.
P301+P312:ПРИ ПРОГЛАТЫВАНИИ: Обратиться за медицинской помощью при плохом самочувствии.
P501:Удалить содержимое/ контейнер на утвержденных станциях утилизации отходов.
Кларитромицин химические свойства, назначение, производство
Описание
Clarithromycin is an acid stable macrolide antibiotic indicated for use in the treatment of skin, urinary and respiratory tract infections. Compared to erythromycin, clarithromycin exhibits the same in vitro activity against conventional pathogens, but is better tolerated by generating less gastrointestinal problems.Химические свойства
Colourless Crystalline NeedlesИспользование
Clarithromycin is a semi-synthetic macrolide antibiotic. Clarithromycin is a derivative of Erythromycin (E650000).Определение
ChEBI: The 6-O-methyl ether of erythromycin A, clarithromycin is a macrolide antibiotic used in the treatment of respiratory-tract, skin and soft-tissue infections. It is also used to eradicate Helicobacter pylori in the treatment f peptic ulcer disease. It prevents bacteria from growing by interfering with their protein synthesis.Антимикробная активность
Activity against susceptible common pathogens is two to four times greater than that of erythromycin A . Most respiratory pathogens, with the exception of H. influenzae, are inhibited at a concentration of ≤0.25 mg/L. It inhibits Mycoplasma pneumoniae at 0.004 mg/L and Mor. catarrhalis at 0.06 mg/L. It is eight times more active than erythromycin A against Legionella spp., C. trachomatis and Ch. pneumoniae. Against anaerobic species, activity is similar to that of erythromycin A. Against H. influenzae the 14-hydroxy metabolite is twice as active as the parent compound.Общее описание
Some of the microbiological properties of clarithromycin appear to be superior to those of erythromycin. It exhibitsgreater potency against M. pneumoniae, Legionellaspp., Chlamydia pneumoniae, H. influenzae, and M. catarrhalisthan does erythromycin. Clarithromycin also hasactivity against unusual pathogens such as Borrelia burgdorferi(the cause of Lyme disease) and the Mycobacteriumavium complex (MAC). Clarithromycin is significantly moreactive than erythromycin against group A streptococci, S.pneumoniae, and the viridans group of streptococci in vivobecause of its superior oral bioavailability. Clarithromycin is,however, more expensive than erythromycin, which must beweighed against its potentially greater effectiveness.Adverse reactions to clarithromycin are rare. The mostcommon complaints relate to GI symptoms, but these seldomrequire discontinuance of therapy. Clarithromycin,like erythromycin, inhibits cytochrome P450 oxidases and,thus, can potentiate the actions of drugs metabolized bythese enzymes.
Clarithromycin occurs as a white crystalline solid that ispractically insoluble in water, sparingly soluble in alcohol,and freely soluble in acetone. It is provided as 250- and 500-mg oral tablets and as granules for the preparation of aqueousoral suspensions containing 25 or 50 mg/mL.
Опасность
Moderately toxic by ingestion. Human sys-temic effects.Фармацевтические приложения
A semisynthetic erythromycin A derivative (6-O- methyl erythromycin A) formulated for oral and intravenous use.Фармакокине?тика
Oral absorption: 55%Cmax 50 mg oral: 0.75 mg/L after 1.7 h
500 mg oral: 1.65 mg/L after 2 h
Terminal half-life: 2.7–3.5 h
Volume of distribution :250 L
Plasma protein binding 80%
absorption and distribution
It is more stable to gastric acid than erythromycin, but internal ketalization between the 9-keto group and the C-12 hydroxyl group has been described resulting in an inactive product: pseudo clarithromycin. It is rapidly absorbed orally and absorption is not affected by food. Concentrations in tonsil and lung tissues exceed the simultaneous plasma level by a factor of two and four, respectively.
Metabolism and excretion
The primary metabolic pathway is N-demethylation of the d-desosamine and stereospecific hydroxylation at the 14- position of the erythronolide A ring. Metabolism to the 14-hydroxy derivative is saturable above 800 mg. Around 20&ndash:40% of the administered dose is eliminated in urine. The apparent elimination half-life of the 14-hydroxy metabolite is around 7 h. The parent compound and its principal metabolite are retained in renal impairment, resulting in long apparent elimination half-lives, exceeding 30 and 45 h, respectively, in patients whose creatinine clearance is less than 30 mL/min.
Побочные эффекты
Clarithromycin is well tolerated, producing little gastrointestinal disturbance and only transient changes in some liver function tests.Кларитромицин запасные части и сырье
Кларитромицин поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия | |
|---|---|---|---|---|---|
| +6596580999 | Singapore | 399 | 58 | ||
| +8615531157085 | China | 8804 | 58 | ||
| +86 13288715578 +8613288715578 |
China | 12825 | 58 | ||
| +86-13131129325 | China | 5887 | 58 | ||
| +86-0571-28186870; +undefined8613073685410 |
China | 1015 | 58 | ||
| +8618092446649 | China | 1143 | 58 | ||
| +86-18239973690 +86-18239973690 |
China | 311 | 58 | ||
| +86-18503288844 +86-18503288844 |
China | 420 | 58 | ||
| +86-(0)57185586718 +86-13336195806 |
China | 29792 | 60 | ||
| +86-0371-55170693 +86-19937530512 |
China | 21632 | 55 |