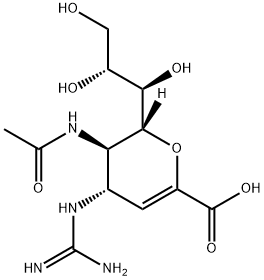
ZANAMIVIR HYDRATE
- русский язык имя
- английское имяZANAMIVIR HYDRATE
- CAS №139110-80-8
- CBNumberCB0292498
- ФормулаC12H20N4O7
- мольный вес332.31
- EINECS691-117-1
- номер MDLMFCD03791324
- файл Mol139110-80-8.mol
| Температура плавления | 2560C (dec.) |
| альфа | D20 +40.9° (c = 0.9 in water) |
| плотность | 1.75±0.1 g/cm3(Predicted) |
| температура хранения | 2-8°C |
| растворимость | H2O: soluble10mg/mL, clear |
| форма | powder |
| пка | 3.82±0.70(Predicted) |
| цвет | white to beige |
| оптическая активность | [α]/D +30 to +40°, c = 1 in H2O |
| Растворимость в воде | Soluble to 5 mM in water |
| Стабильность | Hygroscopic |
| ИнЧИКей | ARAIBEBZBOPLMB-UFGQHTETSA-N |
| Справочник по базе данных CAS | 139110-80-8 |
| Словарь онкологических терминов NCI | Relenza; zanamivir |
| FDA UNII | L6O3XI777I |
| Словарь наркотиков NCI | Relenza |
| Код УВД | J05AH01 |
| UNSPSC Code | 41116107 |
| NACRES | NA.77 |
| Коды опасности | Xn | |||||||||
| Заявления о рисках | 22-36/37/38 | |||||||||
| Заявления о безопасности | 26 | |||||||||
| WGK Германия | 3 | |||||||||
| RTECS | RA9451000 | |||||||||
| кода HS | 2932999000 | |||||||||
| Банк данных об опасных веществах | 139110-80-8(Hazardous Substances Data) | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)

-
сигнальный язык
предупреждение
-
вредная бумага
H315:При попадании на кожу вызывает раздражение.
H319:При попадании в глаза вызывает выраженное раздражение.
H335:Может вызывать раздражение верхних дыхательных путей.
H302:Вредно при проглатывании.
-
оператор предупредительных мер
P301+P312+P330:ПРИ ПРОГЛАТЫВАНИИ: Обратиться за медицинской помощью при плохом самочувствии. Прополоскать рот.
P305+P351+P338:ПРИ ПОПАДАНИИ В ГЛАЗА: Осторожно промыть глаза водой в течение нескольких минут. Снять контактные линзы, если Вы ими пользуетесь и если это легко сделать. Продолжить промывание глаз.
ZANAMIVIR HYDRATE химические свойства, назначение, производство
Описание
Zanamivir was launched as Relenza in Australia for treatment of human influenza A and B virus infections. Zanamivir (4-guanidino-Neu5Ac2en) can be obtained by several similar ways, for instance in seven step synthesis starting from N-acetyI-D-neuraminic acid. Mechanistically, Zanamivir is a potent and specific inhibitor of neuraminidase (or sialidase), a key viral surface glycohydrolase essential for viral replication and disease progression by catalyzing the cleavage of terminal sialic acid residues from the glycoprotein. The in vitro activity of Zanamivir against a wide variety of influenza A and B strains was demonstrated in different model systems; its activity against clinically relevant isolates of influenza virus, with IC50 values ranging from 0.005 to 15 pM was superior to those of amantadine and rimantadine.Химические свойства
Colorless Crystalline SolidИспользование
ZANAMIVIR HYDRATE is a sialic acid analog that inhibits neuraminidase release of newly replicated influenza virus particles. It has been shown to selectively inhibit the growth of influenza A and B viruses in plaque reduction assays with IC50 values ranging from 5 to 14 nM and to directly inhibit influenza A and B virus neuraminidases with IC50 values ranging from 0.6 to 7.9 nM in vitro. The efficacy and tolerabilbity of zanamivir has also been established in clinical trial.[Cayman Chemical]Показания
Zanamivir (Relenza) is a neuraminidase inhibitor with activity against influenza A and B strains. Like oseltamivir, zanamivir is a reversible competitive antagonist of viral neuraminidase. It inhibits the release of progeny virus, causes viral aggregation at the cell surface, and impairs viral movement through respiratory secretions. Resistant variants with hemagglutinin and/or neuraminidase mutations have been produced in vivo; however, clinical resistance to zanamivir is quite rare at present.Приобретенная устойчивость
Resistance is presently uncommon, including strains resistant to oseltamivir. In clinical trials the frequency was no more than 1% of exposed patients.Общее описание
Zanamivir is identical to 2-deoxy-2,3-dehydro-N-acetylneuraminic acid except that itpossesses a guanidino group at position 4 instead of a hydroxylgroup. At positions 119 and 227 of the receptor site,there exist glutamic acid residues. Zanamivir has beenshown to form a salt bridge with the guanidine and Glu-119and a charge transfer interaction with Glu-227. These interactionsincrease the interaction strength with the enzymeand create an excellent competitive inhibitor and an effectiveantiviral agent for influenza types A and B.Human studies have shown that zanamivir is effectivewhen administered before or after exposure to the influenzavirus. If administered before exposure to the virus, the drugreduced viral propagation, infectivity, and disease symptoms.If administered after exposure, the drug reduces propagation,viral titer, and illness. Zanamivir is marketed as a dry powderfor oral inhalation. It is used in adolescents and adults who have been exposed and are symptomatic for not more than 2days. Zanamivir is also indicated for prophylactically treatingfamily members of a person who has developed influenza.
Фармацевтические приложения
A synthetic neuraminidase inhibitor formulated for administration by inhalation.Фармаколо?гия
Zanamivir is generally well tolerated. Bronchospasm and impaired lung function have been reported in some patients taking this medication, but many of these individuals had serious underlying pulmonary disease. Zanamivir should be discontinued if an individual develops bronchospasm or breathing difficulties; treatment and hospitalization may be required. Allergic reactions, including angioedema, have been rarely reported. The efficacy of zanamivir depends upon the proper use of the inhaler device.Фармакокине?тика
Oral bioavailability is poor. After inhalation local respiratory mucosal concentrations greatly exceed those that are inhibitory for influenza A and B replication. The median concentrations in the sputum exceed 1 mg/L 6 h after inhalation and remain detectable for 24 h.Клиническое использование
Treatment of influenza A and B infections in patients over 7 years of age, and prophylaxis of patients ??5 years of ageПобочные эффекты
Most adverse effects are related to the respiratory tree. These include rhinorrhea and, rarely, bronchospasm. Nausea and vomiting have been reported at low incidence.Метаболизм
Zanamirvir is effective when administered via the nasal, intraperitoneal, and intravenous (IV) routes, but it is inactive when given orally. Animal studies have shown 68% recovery of the drug in the urine following intraperitoneal administration, 43% urinary recovery following nasal administration, and only 3% urinary recovery following oral administration. Human data gave results similar to those obtained in animal models. Human efficacy studies with nasal drops or sprays demonstrated that the drug was effective when administered before and after exposure to influenza A or B virus. When given before viral inoculation, the drug reduced viral shedding, infection, and symptoms. When administered beginning at either 26 or 32 hours after inoculation, there was a reduction in shedding, viral titer, and fever.ZANAMIVIR HYDRATE запасные части и сырье
сырьё
- Carbamimidothioic acid, N-acetyl-, methyl ester
- 1H-Pyrazole-3-carboximidamide Hydrochloride
- D-glycero-D-galacto-Non-2-enonic acid, 5-(acetylamino)-4-[(aminoiminomethyl)amino]-2,6-anhydro-3,4,5-trideoxy-, methyl ester, 7,8,9-triacetate
- 4-амино-2-дезокси-2,3-дидегидро-N-ацетилнейраминовая кислота
- Занамивир
1of3
ZANAMIVIR HYDRATE поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия |
|---|---|---|---|---|
| +8615531157085 | China | 8804 | 58 | |
| +86-0371-55170693 +86-19937530512 |
China | 21632 | 55 | |
| 008657128800458; +8615858145714 |
China | 7738 | 55 | |
| +undefined-21-51877795 | China | 32965 | 60 | |
| +86-0371-86658258 +8613203830695 |
China | 29871 | 58 | |
| +8618957127338 | China | 2136 | 58 | |
| 0086-13720134139 | CHINA | 965 | 58 | |
| +1-631-485-4226 | United States | 19553 | 58 | |
| +86-023-6139-8061 +86-86-13650506873 |
China | 39894 | 58 | |
| +8618523575427 | China | 49732 | 58 |