Study on the pharmacological properties of pyrazole
Background
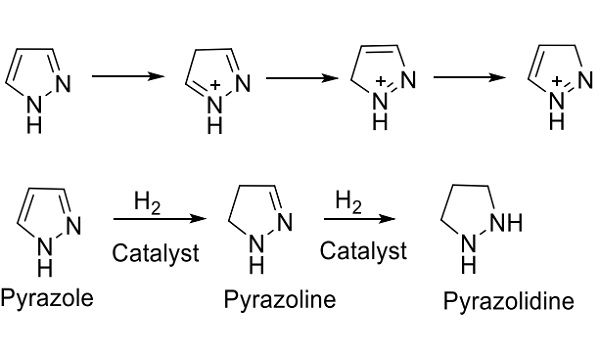
Pyrazole (Figure. 1) are a class of simple aromatic ring organic compounds characterized by a ring structure of 3 carbon atoms and 2 nitrogen atoms that are in adjacent positions also in the unsubstituted parent compound, that are available very rarely in nature.The pyrazole were first isolated from the seeds of watermelon and are classified as alkaloids (occuring confined in few plants) which is the largest groups of chemical arsenals delivered by plants with highly reactive substances with biological activity in low doses that contain nitrogen-usually derived from an amino acid. Synonyms include 1,2-Diazole,1H-Pyrazol and 1H-pyrazole.[1]

Pyrazole is one of the most significant classes of heterocycles, demonstrating a wide range of biological capabilities, such as anticancer, antitumor, anti-AIDS, antimicrobial, antimalarial, and antitubercular. Pyrazole compounds are significant in drug discovery because of their structural variety and range of pharmacological activity, which provide exciting opportunities for the creation of new medicines. In addition to being synthetically accessible and having the possibility for rational design, their relevance as effective molecular scaffolds for medicinal chemistry emerges from their ability to modulate important biological processes implicated in diverse disorders. They have garnered significant interest since these privileged structures are widely found as an active ingredient in commercial medications, such as lonazolac (NSAID), pyrazofurin (anti-cancer), difenamizole (analgesic), deracoxib (NSAID), crizotinib (anti-cancer), tepoxalin (NSAID), fezolamine (antihypertensive), rimonabant (antiobesity), and ruxolitinib (anti-cancer).[2]
1.Pyrazole prevents cisplatin-induced nephrotoxicity in experimental rats.
Cisplatin (a platinum-compound) is a anti-neoplastic drug used in the treatment of various cancers but eventually results in severe adverse effects namely nephrotoxicity or renal disorder through generation of reactive oxygen species (ROS). This biochemical measurements and histopathology analysis investigated a possible protective effect of pyrazole with regards to cisplatin-induced nephrotoxicity in experimental animals. Animals were divided into four groups of six mice each. Group A: normal control, vehicle (1% (w/v) gum acacia in phosphate buffer saline (PBS)). Group B: cisplatin group, vehicle + cisplatin (7.5 mg/kg). Group C: pyrazole (10 mg/kg) + cisplatin and Group D: silymarin (50 mg/kg) + cisplatin. Each vehicle/drug treatment was given daily via intraperitoneal (ip) injection for 10 consecutive days starting from day 1. On group B, C and D cisplatin was given in single dose only on day 5 one hour post drug administration. Animals were allowed till 10th day and on day 11 all four groups animals were anesthetized. Blood samples were collected and serum was isolated for biochemical measurements. The rats were then euthanized by cervical dislocation and their kidney was recovered and then prepared for biochemical measurements and histopathology analyses. Pretreatment with pyrazole prevented nephrotoxicity induced by cisplatin through a protective mechanism that involved reduction of increased oxidative stress by significantly increasing the enzymatic and non enzymatic antioxidant enzymes such as glutathione peroxidase (GPx), glutathione (GSH) and diminishing the lipid peroxidation (LPO). The pretreatment with pyrazole does not affect superoxide dismutase (SOD), catalase (CAT), serum urea and creatinine level during nephrotoxicity when compared to cisplatin-induced group. Moreover, the pyrazole animals shown significant decrease in urine volume and kidney weight when compared with cisplatin-induced group. Histopathological findings reveals the protective efficacy of pyrazole that restores histopathological changes against nephrotoxicity. These analysis will provide a critical evidence that pyrazole could provide a new protective strategy against cisplatin-induced nephrotoxicity.[1]
2. Pyrazole could induce apoptosis in lymphoma cells and mitigate murine solid tumour development
Pyrazole or 1,2-Diazole is a five-membered heteroaromatic ring with two nitrogen atoms which is widely used in pharmacological research and organic synthesis. Several natural and synthetic pyrazole derivatives possess anti-cancer potential and some of them have underwent clinical trials. In this aspect, an investigation into the efficiency of the pyrazole nucleus to inhibit the growth and progression of various cancer cell lines/ experimental tumours would help in giving a better clarity to the anti-cancer behaviour of pyrazole containing drugs. This paper investigates the efficiency of pyrazole against Dalton's Lymphoma Ascites (DLA) cell line. Pyrazole inhibited the growth of DLA cells in vitro by committing them towards apoptosis. In vitro results were consistent in DLA induced murine solid tumour in vivo systems. Drug-treatment improved survival, reduced tumour loads, stabilized body weights and improved the haematological and serum biochemical parameters of DLA solid tumour bearing mice, thereby improving their overall survivability. Drug administration contained the aggravation of solid tumour by targeted downregulation of Cyclin-D1 and Ki-67. In addition, the mRNA expression levels of anti-apoptotic genes, BCL-2 and BCL-XL were downregulated in solid tumours, corroborating the in vitro results that pyrazole encourage apoptotic cell death in DLA cells. The new findings establish pyrazole as a potential anti-cancer drug candidate. The results must encourage future investigations into the efficacy of the drug against various cancer types.[3]
3.Targeting NF-kappaB mediated cell signaling pathway and inflammatory mediators by pyrazole
Lung cancer is the leading cause of cancer deaths globally. The objective of this study was to investigate the effect of pyrazole (1,2-diazole) as an anti-cancer drug on human non-small cell lung carcinoma A549 cells. We attempt to examine the expression level of pro-inflammatory proteins such as TNF-α, NF-κB-p65, MMP-2 and E-Cadherin which are commonly associated with an inflammatory response in epithelial cells and apoptosis in A549 cells. The LPS-induced cytokines and inflammatory mediators include TNF-α, IL-6, iNOS and COX-2 levels in A549 cells and the effect of pyrazole was studied. The present study reveals that, pyrazole inhibits A549 cells by suppressing TNF-α induced MMP-2 expression, thereby inhibiting the nuclear translocation of NF-κB-p65. Pyrazole significantly up-regulate the E-cadherin level and down-regulated MMP-2 expression that could probably preventing A549 cancer cells to invade. The study further substantiated the anti-cancer property of pyrazole by regulating the above mentioned level of LPS-induced cytokines and inflammatory mediators. The observations of the present study open a possibility for the development of an effective therapeutic agent that targets inflammatory and signaling pathway mediators to challenge human non-small cell lung carcinoma.[4]
Conclusion
Pyrazole or 1H-pyrazole, a five-membered 1,2-diazole, is found in several approved drugs and some bioactive natural products. A myriad number of derivatives of this small molecule have been reported in clinical and preclinical studies for the potential treatment of several diseases. The number of drugs containing a pyrazole nucleus has increased significantly in the last 10 years. Some of the best-selling drugs in this class are ibrutinib, ruxolitinib, axitinib, niraparib and baricitinib, and are used to treat different types of cancers; lenacapavir to treat HIV; riociguat to treat pulmonary hypertension; and sildenafil to treat erectile dysfunction. Several aniline-derived pyrazole compounds have been reported as potent antibacterial agents with selective activity against methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci.[5]
References
[1] Prabhu VV, Kannan N, Guruvayoorappan C. 1,2-Diazole prevents cisplatin-induced nephrotoxicity in experimental rats. Pharmacol Rep. 2013;65(4):980-990. doi:10.1016/s1734-1140(13)71079-x
[2] Kumari P, Agrawal N, Mujwar S. Pyrazole Paradigms: Unveiling Synthetic Pathways and Unraveling Anti-Cancer Potential. Med Chem. 2024;20(8):799-817. doi:10.2174/0115734064312273240429110026
[3] Vishnu WK, Abeesh P, Guruvayoorappan C. Pyrazole (1, 2-diazole) induce apoptosis in lymphoma cells by targeting BCL-2 and BCL-XL genes and mitigate murine solid tumour development by regulating cyclin-D1 and Ki-67 expression. Toxicol Appl Pharmacol. 2021;418:115491. doi:10.1016/j.taap.2021.115491
[4] Vinod Prabhu V, Elangovan P, Niranjali Devaraj S, Sakthivel KM. Targeting NF-κB mediated cell signaling pathway and inflammatory mediators by 1,2-diazole in A549 cells in vitro. Biotechnol Rep (Amst). 2021;29:e00594. Published 2021 Jan 28. doi:10.1016/j.btre.2021.e00594
[5] Alam MA. Pyrazole: an emerging privileged scaffold in drug discovery. Future Med Chem. 2023;15(21):2011-2023. doi:10.4155/fmc-2023-0207
Related articles And Qustion
See also
Lastest Price from Pyrazole manufacturers

US $0.00/KG2025-04-21
- CAS:
- 288-13-1
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month

US $25.00/ASSAYS2025-04-21
- CAS:
- 288-13-1
- Min. Order:
- 100ASSAYS
- Purity:
- 99.5%
- Supply Ability:
- 100 mt