Adamantane: Properties and Application of its structure
INTRODUCTION
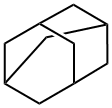
Adamantane (Figure 1) is an affluent natural pharmacophore found in several plants,like Hypericum sinaicum, Hypericum subsessile, Garcinia multiflora, and soon. A compound containing an adamantane skeleton had been isolated from several bacteria and marine invertebrates.[1] Adamantane, a tricyclic C10H16 isomer, is a highly symmetrical, stable tricycle alkane with aunique structure. The adamantane moiety has four tertiary and six secondary carbons and was first searched out in 1933 from crude oil and first synthesized by Prelog and his group.[3]Later, a be tter route was given by Schleyer, centered on the Lewis acid‐catalyzed rearrangement of endo‐trimethylenenorbornane. Schleyer's synthesis opens up new avenues of research on adamantane and its derivatives for developing various pharmaceutical agents and improving the efficacy of known drugs.[2]

PROPERTIES OF ADAMANTANE
The parent molecule, adamantane, exhibits remarkable structural, physical and chemical properties. The molecule is rigid, yet almost unstrained due to the 4 cyclohexyl chairs contained within. As seen in the three-dimensional structure, it presents a spherical topology with a diameter of about 6.4 Å [3] (calculated surface area ~134 Å2, calculated volume ~128 Å3). Compared to a benzene molecule, the adamantane group sweeps out a larger volume and does not present a “Janus” effect (the effective size of benzene depends on the orientation).
Another important difference between adamantane and benzene is the polarizability. Polarizability values have been shown to be related not only to the hydrophobicity but also to biological activities. Unlike benzene (with its quadrupole which interacts with pi-type non-covalent intermolecular forces), the almost spherically symmetric carbon cage of adamantane has an octapole. The value of the static dipole polarizability of adamantane (107.5 e2a°2Eh-1) [4] contrasted with benzene (with a polarizability = 67.48 e2a°2Eh-1) indicates that electrostatic forces between adamantane and its receptor may have a greater effect on binding than previously thought.
The appearance of adamantane as a “grease ball” is apparent in terms of its solubility: it has a high value of the octanol-water partition constant (logP +5.2) which indicates it is completely insoluble in water. In addition, its high symmetry (Td) influences the high melting point (269 °C,unusual for a hydrocarbon) and crystallinity.
Since the molecule only contains aliphatic carbons and hydrogen atoms, it is unusually inert. The hydrogens are tertiary and secondary (no primary or methyl) which restricts the level of reactivity (e.g. oxidation). Moreover, the robust tricyclic system inhibits elimination or transposition reactions that may occur in other reactive systems. However,many mono, di, tri and tetra-derivatives can be formed (usually at the bridgehead positions) which provides an unique tetrahedral core.[5]
The adamantane moiety has attracted significant attention since its discovery in 1933 due to its remarkable structural, chemical, and medicinal properties. This molecule has a notable impact in the therapeutic field because of its “add‐on” lipophilicity to any pharmacophoric moieties. As in the case of molecular hybridization, in which one pharmacophore is attached to another one(s) with a probability of increasing the biological activity, adding an adamantane unit improves the absorption distribution, metabolism and excretion properties of the resultant hybrid molecule.
APPLICATION OF ADAMANTANE STRUCTURE
Adamantane backbone can be transformed at various positions to yield multifunctional adamantane derivatives with improved properties. Several substitution reactions of adamantane were reported in 1961.However, chemists are more interested in modifying the bridgehead positions mainly because of ease inselective C–H activation. Adamantane‐based structurally modified compounds were found to have several applications in various fields including supramolecular chemistry, catalysis, surface functionalization, biocompatible surface tailoring, materials science, and so on. The first adamantane‐based drug was amantadine (1‐amino adamantane), which displayed potent anti‐influenza and anti‐Parkinson activity. Further, studies led to the discoveries of potent antiviral drugs rimantadine and tromantadine. Several commercially available drugs containing adamantane moiety are amantadine, rimantadine, tromantadine,adapromine (antiviral), adapalene (for acne treatment), vildagliptin,saxagliptin (antidiabetic), bromantane (anxiolytic), memantine (anti‐Alzheimer), and SQ109 (anti‐tuberculosis [TB]).[2]
Good hydrophobicity, lipophilicity as well as well‐defined three‐dimensional structure may be responsible for the transport of adamantane‐based molecules through biological membranes.This molecule has a remarkable impact in the medicinal field because of its “add‐on” lipophilicity to any pharmacophoric moieties, which allows adamantane‐based compounds to pass through cell membranes. As in the case of molecular hybridization, in which one pharmacophore is attached to another one(s) with a probability of increasing biological activity, adding an adamantane unit completely modifies the absorption distribution, metabolism and excretion properties of the resultant hybrid molecule. As reported in the literature, the above properties help improve some known leads for pharmacokinetic and pharmacodynamic properties. Adamantane‐based compounds are reported to possess a plethora of biological activities including antiviral, anticancer, antimalarial, antimicrobial, and inhibitors of various enzymes.[2]
CONCLUSION
Adamantane is considered a “lipophilic bullet,” which shows an “add-on”behavior toward lipophilicity when attached to any pharmacophoric moieties. This molecule also positively impacts the ADME properties of the resultant hybrid molecule, thereby changing the pharmacokinetics and pharmacodynamics. Further, many more multivalent architectures with an adamantane scaffold as a rigid core can be constructed. The diverse biological activities illustrated by adamantane derivatives have influenced the research on this potent scaffold.
References
[1] Fort R. C. , Schleyer P. V. R .Adamantane: Consequences of the Diamondoid Structure[J].Chemical Reviews, 1964, 64(3):277-300.
[2] Ragshaniya A., Kumar V., Tittal R.K., Lal K. Nascent pharmacological advancement in adamantane derivatives. Arch Pharm (Weinheim). 2024;357(3):e2300595.
[3] Morel-Desrosiers, N., Morel, J.-P. Standard molar enthalpies, volumes,and heat capacities of adamantane in cyclohexane, n-hexane,and carbon tetrachloride. Interpretation using the scaled-particletheory. J. Solution Chem. 1979, 8: 579-592.
[4] Maroulis, G., Xenides, D., Hohm, U., Loose, A. Dipole, dipolequadrupole,and dipole-octopole polarizability of adamantane,C10H16, from refractive index measurements, depolarized collision induced light scattering, conventional ab initio and density functional theory calculations J. Chem. Phys. 2001, 115:7957-7967.
[5] Lamoureux G., Artavia G. Use of the adamantane structure in medicinal chemistry. Curr Med Chem. 2010;17(26):2967-2978.
See also
Lastest Price from Adamantane manufacturers

US $10.00/ASSAYS2025-08-22
- CAS:
- 281-23-2
- Min. Order:
- 1ASSAYS
- Purity:
- 99%
- Supply Ability:
- 1 ton

US $5.00-0.50/KG2025-05-30
- CAS:
- 281-23-2
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS