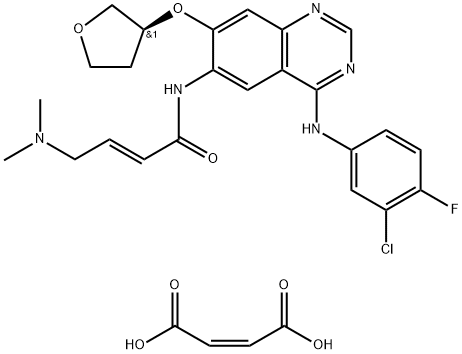
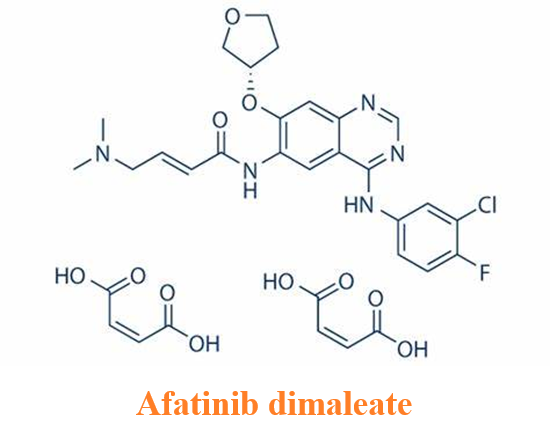
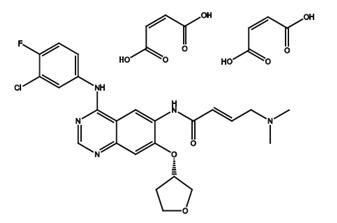
Side effects of afatinib dimaleate
Afatinib dimaleate, the dimaleate form of afatinib, is an orally bioavailable anilinoquinazoline derivative that is an inhibitor of the epidermal growth factor receptor (ErbB; EGFR) family of receptor tyrosine kinases (RTKs) with antitumor activity. Afatinib is sold under the brand name Gilotrif, among others, and is used to treat non-small cell lung cancer (NSCLC).

Mechanism of Action
After administration, afatinib selectively and irreversibly binds to and inhibits epidermal growth factor receptors 1 (ErbB1; EGFR), 2 (ErbB2; HER2), and 4 (ErbB4; HER4), as well as certain EGFR mutants, including those caused by EGFR exon 19 deletion mutations or exon 21 (L858R) mutations. This may result in inhibition of tumor growth and angiogenesis in tumor cells that overexpress these RTKs. In addition, afatinib inhibits the EGFR T790M gatekeeper mutation, which confers resistance to treatment with first-generation EGFR inhibitors. EGFR, HER2, and HER4 are RTKs that belong to the EGFR superfamily; they play a major role in tumor cell proliferation and tumor angiogenesis and are overexpressed in many cancer cell types.
Side Effects
The side effects that may occur in clinical use of Afatinib dimaleate (Gilotrif) mainly include:
Gastrointestinal reactions (common): loss of appetite, diarrhea, stomatitis, nausea, vomiting and constipation
Skin and subcutaneous reactions: rash, acne-like dermatitis, itching, dry skin and hair loss
Infections: cheilitis, paronychia, nasopharyngitis, cystitis and upper respiratory tract infection
Metabolic and nutritional disorders: hypokalemia
Nervous system disorders: headache, dizziness, insomnia and taste disorders
Eye diseases: conjunctivitis
Others: fatigue, fever, cough and weight loss, etc.
In addition, abnormal liver function tests (including elevated alanine aminotransferase [ALT] and aspartate aminotransferase [AST]) were observed in patients treated with GIOTRIF 40 mg. These increases were mainly temporary and did not lead to discontinuation of treatment. Grade 2 (>2.5 to 5.0 times ULN [upper limit of normal]) ALT elevations occurred in 7.9% and 3.6% of patients treated with GIOTRIF or chemotherapy, respectively.
References:
[1] Afatinib Dimaleate[J]. Definitions, 2020, 41 1. DOI:10.32388/e26ogb.
Related articles And Qustion
See also
Lastest Price from Afatinib dimaleate manufacturers

US $0.00/g2025-04-21
- CAS:
- 850140-73-7
- Min. Order:
- 1g
- Purity:
- 99%min
- Supply Ability:
- 1000g
US $0.00-0.00/g2025-04-20
- CAS:
- 850140-73-7
- Min. Order:
- 10g
- Purity:
- 99% HPLC
- Supply Ability:
- 10000