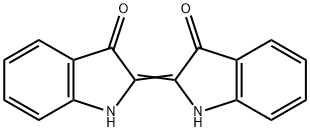
Production Methods of Indigo
Indigo, or indigotin, is a dyestuff originally extracted from the varieties of the indigo and woad plants. Indigo was known throughout the ancient world for its ability to color fabrics a deep blue. Egyptian artifacts suggest that indigo was employed as early as 1600 B.C. and it has been found in Africa, India, Indonesia, and China.

The raw materials used in the natural production of indigo are leaves from a variety of plant species including indigo, woad, and polygonum. Only the leaves are used since they contain the greatest concentration of dye molecules. In the synthetic process, a number of chemicals are employed as described below.Originally extracted from plants, today indigo is synthetically produced on an industrial scale. Indigo has the following production methods.
Natural extraction
Plant extraction of indigo requires several steps because the dye itself does not actually exist in nature. The chemical found in plant leaves is really indican, a precursor to indigo. The ancient process to extract indican from plant leaves and convert it to indigo has remained unchanged for thousands of years. In this process, a series of tanks are arranged in a step wise fashion. The upper-most tank is a fermentation vessel into which the freshly cut plants are placed. An enzyme known as indimulsin is added to hydrolyze, or break down, the indican into indoxyl and glucose. During this process carbon dioxide is given off and the broth in the tank turns a murky yellow.
After about 14 hours, the resulting liquid is drained into a second tank. Here, the indoxyl-rich mixture is stirred with paddles to mix it with air. This allows the air to oxidize the indoxyl to indigotin, which settles to the bottom of the tank. The upper layer of liquid is siphoned away and the settled pigment is transferred to a third tank where it is heated to stop the fermentation process. The resultant mixture is filtered to remove impurities and dried to form a thick paste.
Historically, the Japanese have used another method which involves extracting indigo from the polygonum plant. In this process the plant is mixed with wheat husk powder, limestone powder, lye ash, and sake. The mixture is allowed to ferment for about one week to form the dye pigment which is called sukumo.
Synthetic production
A variety of synthetic chemical processes have been used to produce indigo. All these processes involve combining a series of chemical reactants under controlled conditions. The reactants undergo a series of reactions which result in the formation of the indigo molecule. A number of other chemical byproducts are also produced in this reaction.
These synthesis reactions are conducted in large stainless steel or glass reaction vessels. These vessels are equipped with jackets to allow steam or cold water to flow around the batch as the reactions progress. Because of the complexity of these chemical processes, the dye is usually made in batch quantities. There are, however, a few methods invented by the Germans for continuous process manufacturing.
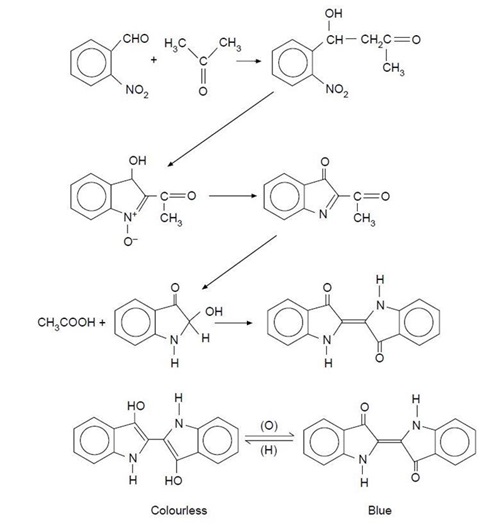
The first commercial method of producing indigo was based on Heumann's work. In this method, N-phenylglycine is treated with alkali to produce indoxyl, which can be converted to indigotin by contact with air. However, the amount of dye yielded by this process is very low. Another, more efficient, synthesis route utilizes anthranilic acid. This process was popular with major manufacturers, such as BASF and Hoechst, for over 30 years. A variation of this method (which has become widely used) involves the reaction of aniline, formaldehyde, and hydrogen cyanide to form phenylglycinonitrile. This material is then hydrolyzed to yield phenylglycine which is then converted to indigotin. Currently, a method which uses sodamide with alkali to convert phenylglycine to indoxyl. Sodamide reacts with excess water, thus lowering the overall reaction temperature from almost 570°F (300°C) to 392°F (200°C). This results in a much more efficient reaction process.After the chemical reaction process is complete, the finished dye must be washed to remove impurities and then dried. The dried powder can be packed in drums or reconstituted with water to form a 20% solution and filled in pails.
References
[1] http://www.madehow.com/Volume-6/Indigo.html
[2] https://thekindcraft.com/the-process-indigo-from-plant-to-paste
[3] Nicholas Wenner. The Production of Indigo Dye from Plants. Fibershed[J]. 2017(10):2-4
You may like
Related articles And Qustion
See also
Lastest Price from Indigo manufacturers

US $10.00/KG2025-04-21
- CAS:
- 482-89-3
- Min. Order:
- 100KG
- Purity:
- 99%
- Supply Ability:
- 100 mt

US $79.00-38.00/kg2025-04-21
- CAS:
- 482-89-3
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20ton