NAD+:Introduction;Biosynthesis;Metabolism in aging and disease
Introduction
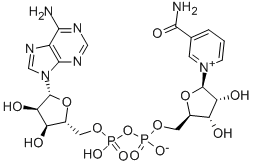
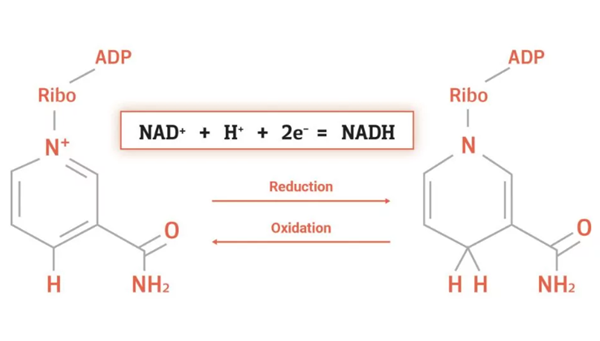
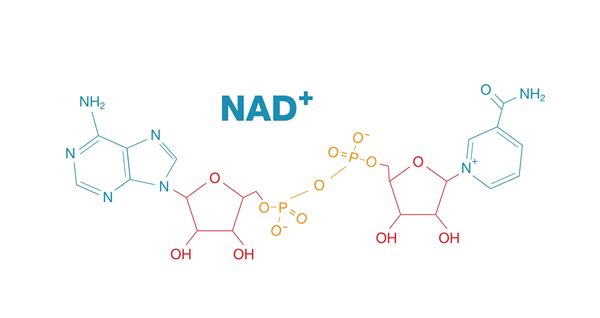
NAD+ is a coenzyme consisting of ribosyl nicotinamide 5'-diphosphate coupled to adenosine 5'-phosphate via a pyrophosphate bond. The first identified function of nicotinamide adenine dinucleotide (NAD+) was its ability to reversibly bind hydride ions, which are formally defined as anions of hydrogen (H−).
This chemical property is known to play a crucial role in cellular metabolism, where NAD+serves as an electron acceptor in oxidation–reduction (redox) reactions during the breakdown of energetic substrates (e.g, glucose, fatty acids), thus generating NADH as reducing equivalent. Apart from these fundamental redox properties, numerous further processes crucial for cell vitality are dependent on NAD+. For instance, NAD+serves as a substrate for NAD+-consuming enzymes, such as sirtuins (SIRTs), poly(ADP-ribose) polymerases (PARPs), and ectoenzymes like cluster of differentiation (CD)38, CD157, or the recently discovered sterile alpha and toll interleukin receptor motif-containing protein 1 (SARM1). Despite the apparent structural resemblance, NADP+, the phosphorylated form of NAD+, has rather distinct functions in cellular metabolism. NADP+ is reduced to NADPH in the pentose phosphate pathway and serves as a redox partner in the reductive biosynthesis of fatty acids, cholesterol, and steroid hormones, hepatic detoxification of xenobiotics, and oxidative defenses.
NAD+ Biosynthesis
NAD+ biosynthesis can broadly be divided into the kynurenine pathway, the Preiss-Handler pathway, and a salvage pathway. While these pathways all generate NAD+ from different nutritional precursors, the salvage pathway additionally recycles NAD+ from nicotinamide—a cleavage product arising from the activity of NAD+-consuming enzymes(Figure 1).
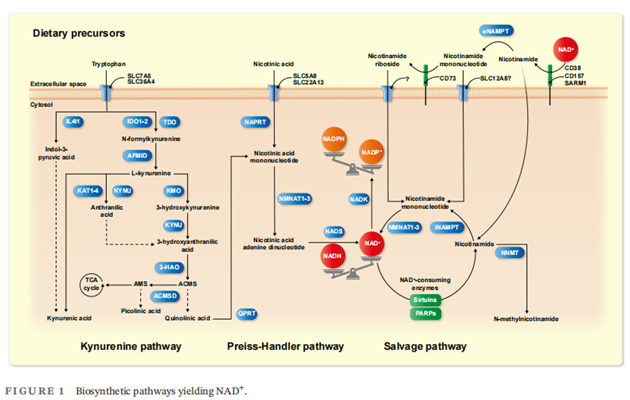
Intracellular levels of NAD+ are held constant via three distinct pathways using the dietary precursors tryptophan, nicotinic acid, nicotinamide riboside, nicotinamide mononucleotide, and nicotinamide. The kynurenine pathway depicts the major catabolic route of the essential amino acid tryptophan, generating quinolinic acid (QA) as one of its end products. QA then converges with the PreissHandler pathway, ultimately yielding NAD+. The salvage pathway either recycles NAD+ from nicotinamide after it has been cleaved by NAD+-consuming enzymes or incorporates dietary precursors. Note, that the illustration above serves as a schematic overview of NAD+- synthesizing pathways. For tissue-specific and subcellular expression patterns of the corresponding enzymes please refer to Ref. 15. 3-HAO, 3-Hydroxyanthranilate 3,4-dioxygenase; ACMS, α-amino-β-carboxymuconate ε-semialdehyde; ACMSD, aminocarboxymuconate semialdehyde decarboxylase; AFMID, arylformamidase; AMS, 2-aminomuconic-6-semialdehyde; IDO, indolamine 2,3-dioxygenase; IL4I1, interleukin 4 induced 1; iNAMPT and eNAMPT, intracellular and extracellular nicotinamide phosphoribosyltransferase; KAT, kynurenine aminotransferase; KMO, kynurenin-3-monooxygenase; KYNU, kynureninase; NADK, NAD kinase; NADS, NAD synthase; NAPRT, nicotinate phosphoribosyltransferase; NMNAT, nicotinamide mononucleotide adenylyltransferase; NNMT, nicotinamide N-methyltransferase; NRK, nicotinamide riboside kinase; PARPs, poly(ADP-ribose) polymerases; QPRT, quinolinate phosphoribosyltransferase; SARM1, sterile alpha and toll interleukin receptor motif–containing protein 1; SLC, solute carrier; TCA, tricarboxylic acid; TDO, tryptophan 2,3-dioxygenase.
NAD+ metabolism in aging and disease
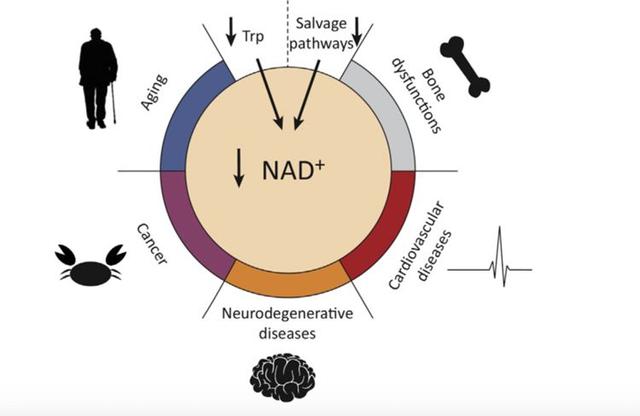
Over the last decades alterations in NAD+ metabolism have been revealed in multiple age-associated diseases, including neoplastic, metabolic, and neurodegenerative diseases, as well as aging per se. Depending on the investigated population (rodents vs. humans) and tissue type, the NAD+ levels of aged subjects were reported to range from 15% to 90% of normal tissue levels. Current hypotheses for this age-dependent decline revolve around the frequently formulated “hallmarks of ageing”, including loss of proteostasis and genomic stability.
NAD+ biosynthesis enzymes become dysfunctional during aging, resulting in decreased NAD+ levels. Additionally, the age-associated accumulation of DNA damage is thought to increase PARP activity, reinforcing the cellular decline of NAD+. 29 This renders NAD+-dependent processes such as energy metabolism and DNA repair ineffective, leading to age-related pathophysiology and tissue degeneration. Despite overall claims of NAD+ decline with age the scientific evidence is rather limited and often restricted to certain tissue types.
References:
[1] DAVID WALZIK. Tissue-specific effects of exercise as NAD+-boosting strategy: Current knowledge and future perspectives[J]. Acta Physiologica, 2023, 237 3. DOI:10.1111/apha.13921.
Related articles And Qustion
See also
Lastest Price from β-Nicotinamide adenine dinucleotide manufacturers

US $10.00-7.00/VIAL2025-11-21
- CAS:
- Min. Order:
- 10VIAL
- Purity:
- 99.9
- Supply Ability:
- 20000

US $50.00-10.00/Box2025-11-12
- CAS:
- Min. Order:
- 1Box
- Purity:
- 99.99%
- Supply Ability:
- 3000vials