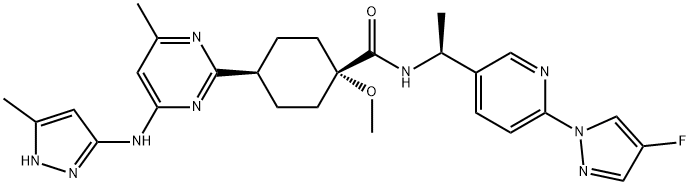
Does Pralsetinib need to be reduced in patients with liver impairment?
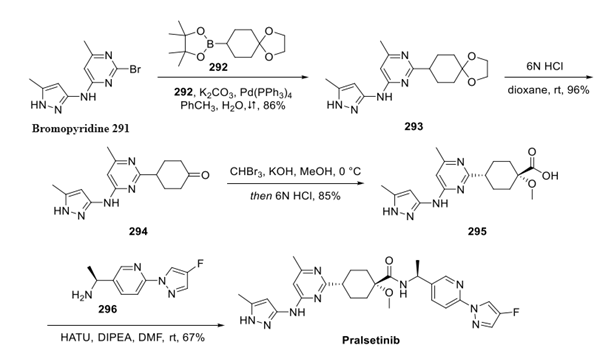
No. Pralsetinib is a kinase inhibitor used to treat metastatic rearranged during transfection (RET) fusion-positive non-small cell lung cancer. Pralsetinib is primarily cleared by the liver, so hepatic impairment (HI) may alter its pharmacokinetics (PK). However, studies have shown that mild HI has minimal effects on the PK of pralsetinib. In addition, based on the Child–Pugh classification, the systemic exposure (area under the plasma concentration-time curve from time 0 to infinity [AUC0–∞]) to pralsetinib in subjects with moderate and severe HI was similar to that in subjects with normal hepatic function, with AUC0–∞ geometric mean ratios (GMRs) of 1.12 and 0.858, respectively. Results based on the NCI-ODWG classification criteria were comparable. According to the NCI-ODWG, the AUC0–∞ GMRs for subjects with moderate and severe HI compared with subjects with normal hepatic function were 1.22 and 0.858, respectively. These results suggest that moderate and severe hepatic impairment do not significantly affect the exposure of pralsetinib, and therefore no dose adjustment is required in this population.

References:
[1] CHEUNG K W K, TANG Y, ANDERS D, et al. Exploring the Impact of Hepatic Impairment on Pralsetinib Pharmacokinetics[J]. Pharmaceutics, 2024, 7 1. DOI:10.3390/pharmaceutics16040564.
You may like
Related articles And Qustion
See also
Lastest Price from Pralsetinib manufacturers
US $0.00-0.00/mg2025-04-18
- CAS:
- 2097132-94-8
- Min. Order:
- 10mg
- Purity:
- 98
- Supply Ability:
- 10000000

US $0.00/g2025-01-13
- CAS:
- 2097132-94-8
- Min. Order:
- 1g
- Purity:
- More Than 99%
- Supply Ability:
- 100kg/Month