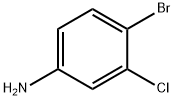
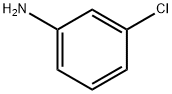
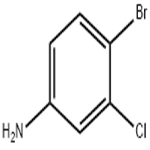
General method: A solution was prepared by dissolving m-chloroaniline (2a or 2b) in DMF as starting material. Subsequently, another solution was prepared by dissolving NBS (92 mmol, 1.0 eq.) in 100 ml DMF. The DMF solution of m-chloroaniline was added dropwise to the DMF solution of NBS at room temperature. After the dropwise addition, the reaction mixture was stirred continuously for 3 hours at room temperature. After completion of the reaction, the reaction mixture was diluted with 500 ml of ethyl acetate and washed with 2 x 150 ml of brine. The organic phase was separated, dried with anhydrous Na2SO4, filtered and concentrated to give 4-bromo-3-chloroaniline (3a or 3b) as a brown solid in 90-92% yield. In another experiment, m-chloroaniline (2a or 2b, 0.8 mmol) was dissolved in a solvent of choice (1 ml) and a solution of NBS (0.8 mmol) in the same solvent (1 ml) was added in batches at room temperature. The reaction progression was monitored by LC-MS/MS and a final mixture containing traces of the dibrominated product was obtained.
[1] Medicinal Chemistry Research, 2016, vol. 25, # 4, p. 539 - 552
[2] Patent: CN107089919, 2017, A. Location in patent: Paragraph 0044; 0045; 0046; 0047
[3] Synthetic Communications, 2009, vol. 39, # 10, p. 1817 - 1824