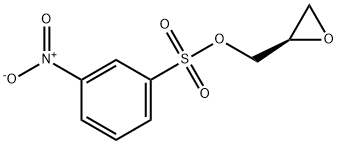
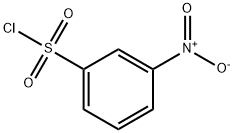
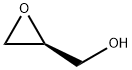
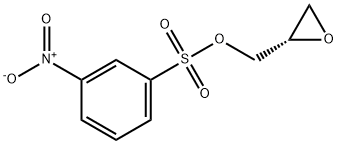
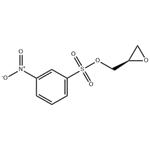
Preparation of Compound 3: To an anhydrous dichloromethane (10 mL) and triethylamine (1.5 mL, 10 mmol) solution of (S)-oxiran-2-ylmethanol (220 mg, 3.0 mmol) that had been cooled to 0 °C (ice bath), 3-nitrobenzenesulfonyl chloride (730 mg, 3.3 mmol) was added in batches. The resulting brown solution was stirred at 0 °C for 1 h, followed by slow warming to room temperature. The progress of the reaction was monitored by TLC every 15 min until (S)-oxiran-2-ylmethanol was completely consumed. Upon completion of the reaction, the reaction was quenched by the addition of crushed ice (1-2 g). The reaction mixture was transferred to a dispensing funnel and partitioned with dichloromethane (20 mL) and water (10 mL). The organic layer was washed sequentially with saturated aqueous NaHCO3 (10 mL x 3) and brine (10 mL x 3), followed by drying with anhydrous Na2SO4. After filtration and concentration, the residue was purified by fast column chromatography to afford (S)-(+)-m-nitrobenzenesulfonic acid glycidyl ester (Compound 3) as a brown solid (710 mg, 92%).1H NMR (400 MHz, CDCl3) δ 8.77 (s, 1H), 8.54 (d, J=8.1 Hz, 1H), 8.28 (d, J=7.9 Hz , 1H), 4.50 (d, J=11.5Hz, 1H), 4.04 (dd, J1=4.9Hz, J2=1.5Hz, 1H), 3.23 (d, J=1.8Hz, 1H), 2.85 (dd, J1=4.0Hz, J2=2.0Hz, 1H), 2.64 (dd, J1=2.4Hz, J2=2.0Hz, 1H) 1H); 13C NMR (100 MHz, CDCl3) δ 148.19, 137.92, 133.32, 130.81, 128.39, 123.16, 71.69, 48.64, 44.42.