p-Toluic Acid is used as a reagent in the synthesis of several organic compounds including that of 2-aryl-1,3,4-oxadiazole derivatives which are potential antibacterial agents. Also used in the synthesis of 4-(bromomethyl) benzoic acid.
colourless crystals or white crystalline powder. Slightly soluble in water; soluble in alcohol and ether. Combustible.
Intermediates of Liquid Crystals
p-Toluic Acid is used as a reagent in the synthesis of several organic compounds including that of 2-aryl-1,3,4-oxadiazole derivatives which are potential antibacterial agents. Also used in the synthesis of 4-(bromomethyl) benzoic acid.
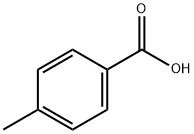
ChEBI: A methylbenzoic acid in which the methyl substituent is located at position 4.
Fine dust dispensed in air in sufficient concentrations, and in the presence of an ignition source is a potential dust explosion hazard. . Insoluble in water.
p-Toluic acid is a carboxylic acid. Carboxylic acids donate hydrogen ions if a base is present to accept them. They react in this way with all bases, both organic (for example, the amines) and inorganic. Their reactions with bases, called "neutralizations", are accompanied by the evolution of substantial amounts of heat. Neutralization between an acid and a base produces water plus a salt. Carboxylic acids with six or fewer carbon atoms are freely or moderately soluble in water; those with more than six carbons are slightly soluble in water. Soluble carboxylic acid dissociate to an extent in water to yield hydrogen ions. The pH of solutions of carboxylic acids is therefore less than 7.0. Many insoluble carboxylic acids react rapidly with aqueous solutions containing a chemical base and dissolve as the neutralization generates a soluble salt. Carboxylic acids in aqueous solution and liquid or molten carboxylic acids can react with active metals to form gaseous hydrogen and a metal salt. Such reactions occur in principle for solid carboxylic acids as well, but are slow if the solid acid remains dry. Even "insoluble" carboxylic acids may absorb enough water from the air and dissolve sufficiently in p-Toluic acid to corrode or dissolve iron, steel, and aluminum parts and containers. Carboxylic acids, like other acids, react with cyanide salts to generate gaseous hydrogen cyanide. The reaction is slower for dry, solid carboxylic acids. Insoluble carboxylic acids react with solutions of cyanides to cause the release of gaseous hydrogen cyanide. Flammable and/or toxic gases and heat are generated by the reaction of carboxylic acids with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. Carboxylic acids, especially in aqueous solution, also react with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), to generate flammable and/or toxic gases and heat. Their reaction with carbonates and bicarbonates generates a harmless gas (carbon dioxide) but still heat. Like other organic compounds, carboxylic acids can be oxidized by strong oxidizing agents and reduced by strong reducing agents. These reactions generate heat. A wide variety of products is possible. Like other acids, carboxylic acids may initiate polymerization reactions; like other acids, they often catalyze (increase the rate of) chemical reactions. p-Toluic acid is incompatible with strong oxidizers. p-Toluic acid is also incompatible with strong bases. .
Flash point data for p-Toluic acid are not available; however, p-Toluic acid is probably combustible.
Flammability and Explosibility
Not classified
Crystallise the acid from water, water/EtOH (1:1), MeOH/water or *benzene. [Beilstein 9 IV 1724.] Aromatic acid impurities (to <0.05%) can be removed via the (±)-�-methylbenzylamine salt as described for 2,4-dichlorobenzoic acid [Ley & Yates Organic Process Research & Development 12 120 2008]. The S-benzylisothiuronium salt has m 164o (from aqueous EtOH).