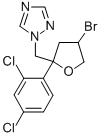
BROMUCONAZOLE
- Product NameBROMUCONAZOLE
- CAS116255-48-2
- MFC13H12BrCl2N3O
- MW377.06
- EINECS
- MOL File116255-48-2.mol
Chemical Properties
| Melting point | 84℃ |
| Boiling point | 504.3±60.0 °C(Predicted) |
| Density | 1.71 |
| vapor pressure | 50 mg 1-1 |
| refractive index | 1.6110 (estimate) |
| storage temp. | -20°C |
| solubility | Chloroform (Slightly), Methanol (Slightly) |
| pka | 2.75±0.10(Predicted) |
| BRN | 8393967 |
| Henry's Law Constant | 3.3×104 mol/(m3Pa) at 25℃, Duchowicz et al. (2020) |
| Major Application | agriculture environmental |
| InChI | 1S/C13H12BrCl2N3O/c14-9-4-13(20-5-9,6-19-8-17-7-18-19)11-2-1-10(15)3-12(11)16/h1-3,7-9H,4-6H2 |
| InChIKey | HJJVPARKXDDIQD-UHFFFAOYSA-N |
| SMILES | Clc1ccc(c(Cl)c1)C2(CC(Br)CO2)Cn3cncn3 |
| LogP | 3.240 |
| EPA Substance Registry System | Bromuconazole (116255-48-2) |
Safety Information
| Hazard Codes | Xn |
| Risk Statements | 22 |
| Safety Statements | 36 |
| RIDADR | UN 3077 9 / PGIII |
| WGK Germany | 3 |
| RTECS | XZ4130000 |
| HazardClass | 6.1 |
| PackingGroup | III |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Acute Tox. 4 Oral Aquatic Acute 1 Aquatic Chronic 1 Repr. 2 |
| Hazardous Substances Data | 116255-48-2(Hazardous Substances Data) |
| Toxicity | LD50 orally in rats, mice: 365, 1151 mg/kg; LD50 dermally in rats: >2000 mg/kg; LD50 by inhalation in rabbits: >5 mg/l; LC50(96 hr) in rainbow trout, bluegill sunfish (mg/l): 1.7, 3.1 (Pepin) |