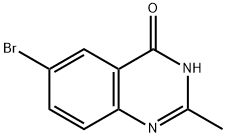
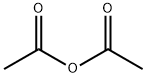
The general procedure for the synthesis of 6-bromo-2-methylquinazolin-4(3H)-one from ethanoic anhydride and 2-amino-5-bromobenzoic acid was as follows: first, 2-amino-5-bromobenzoic acid was refluxed for 1 h in liquid ammonia (10 mL/g). Subsequently, the reaction solution was concentrated to a smaller volume and the residue was mixed with acetic anhydride (2 mL/g) and refluxing was continued for 4 hours. Upon completion of the reaction, water was added to the mixture and boiled for 30 minutes, followed by dropwise addition of 20% sodium hydroxide solution until a clarified solution was formed. Next, ammonium carbonate was added to the hot solution until a precipitate was formed. The mixture was allowed to stand at 4°C for 16 hours, after which the precipitate was collected by filtration, washed with water and finally dried under vacuum to give the target product in about 60% yield.